-
Services
-
Featured Strengths
-
In Vitro ADME
-
Rodent In Vivo PK
-
Metabolite Identification
-
Bioanalysis
-
Case Study

Services
WuXi AppTec DMPK New Jersey traces its roots to XenoBiotic Laboratories (XBL), founded in 1987 and acquired by WuXi AppTec in 2014. Today it operates as a WuXi AppTec subsidiary located in Cranbury, New Jersey, providing DMPK services. We serve as a trusted strategic partner for global pharmaceutical and biotech companies, and our team of experts offers comprehensive solutions to complex R&D challenges. We combine quality, capacity and cross-disciplinary coordination to turn your drug development goals into reality, providing end-to-end support from discovery through IND and NDA.
-

 In Vitro ADME
In Vitro ADME

Screening, IND Enabling, and Definitive in vitro ADME and DDI Assays
-

 Rodent In Vivo PK
Rodent In Vivo PK

Discovery and Preclinical Development Radiolabeled and Non-Radiolabeled Rodent PK/PD, Mass Balance, and QWBA
-

 Metabolite Identification
Metabolite Identification

Discovery, IND-Enabling (Including Radiolabeled), Preclinical & Clinical Development Met ID Service
-

 Bioanalysis
Bioanalysis

LC-MS/MS, Immunochemistry (LBA), DNA/RNA Analysis, Cell-Based Assays, and Clinical Chemistry & Hematology

Featured Strengths
-
Local for Local
Efficient US-Based Services for Local Compounds/Samples
Responsive Local Contact & Easy On-Site Visits
Clinical DMPK/Human AME, Specialized Cells/Matrices/Reagents/Strains Available
-
Global for Local
Global Resources for Better Local Services
US-Based Compound & Sample Shipping Hub for Global Service Access
Unified Global QC Standards and Protocols
In Vitro ADME
-
Drug Discovery
CYP inhibition screening
PXR/AHR activation screening
Physicochemical properties
Plasma protein binding (HTD/RED/UC methods)
Blood-to-plasma Ratio
Plasma/microsomal/S9/hepatocyte stability
Cell permeability screening assay uses Caco-2 and MDR1-MDCK cells incubations in transwell plates
Additional assays available upon request
-
Preclinical Development (IND Stage)
CYP direct and time dependent inhibition (full panels)(7-9 CYPs) – direct (DI) and time-dependent (TDI): IC50, Ki, reversibility, kinact/KI
UGT inhibition (1A1, 1A4, 1A9, 2B7 and 2B15)
Phenotyping CYP, UGT, FMO, MAO, AO, XO, CES, SULT, ALDH, ADH
CYP induction mRNA and activity based - CYP1A2, 2B6, 3A4, 2C8, 2C9, 2C19
PgP (MDR1) and BCRP inhibition and substrate
-
Clinical Development (NDA Stage)
Full panels of FDA/EMA/ICH M12 transporter
Substrate and inhibition
Efflux transporters
Cell based: MDCKII-MDR1, MDCKII- BCRP, and Caco-2 (monolayer)
Vesicles: BCRP, MDR1, BSEP, MRP2, MRP3, MRP4
Uptake transporters
Cell Based: OAT1, OAT3, OATP1B1, OATP1B3, OATP2B1, OCT1, OCT2, PEPT1, PEPT2, MATE1, MATE2-K, NTCP, ASBTDrug–drug interaction and mechanism-based follow-up study supporting clinical development
Ex vivo assessment of human plasma protein binding
Rodent In Vivo PK
-
Discovery PK Service
Service type: single/multiple dose PK, Tissue distribution, Brain penetration, MTD, etc.
Naïve, knockout, transgenic, immunodeficient and other commercially available rodent strains
-
Radiolabeled PK Study
Radioisotope: 14C & 3H
Service type: Mass balance, bile excretion, Radio-PK, Quantitative Whole-body Autoradiography (QWBA), Quantitative Tissue Dissection (QTD)
-
Administration and Sample Collection
IV, PO, IP, ICV, IN, IT, sub-Q (including osmotic pump and slow-release pellet), and oral film
Serial bleeding up to 8 time points in 24 hours
70+ tissue and tissue fluid collection capabilities, specializing in CNS and ophthalmology
-
Instruments and software
KOPF Model 940 small animal stereotaxic instrument
Revvity Liquid Scintillation Counters and Sample Oxidizer
Leica/Vibratome Microtomes
Typhoon Phosphor Imager
Glass stability chambers
AIDA-image analysis software
DEBRA LIMS Radioactive ADME software
WinNonlin (Phoenix) PK/PD modeling software
Bioanalysis
-
-
LCMS
Triple QuadTM (API6500, API6500+, Xevo TQs, Xevo, TQ Absolute)
High resolution API QTOF6600, UPLC-PDA-FLD
-
Immunochemistry
MESOTM QuickPlex SQ120 & Sector S600SpectraMax i3x & iD5
EnVision 2105
ELISpot
Quanterix HD-XNanodropTM One
LSRFortessaTM X-20 & FACSVerse
-
DNA & RNA Detection & Analysis
Real-time PCR 7500, QuantStudio 6 & 7 Pro
PROFLEX PCR System
QX600 (ddPCR)
KingFisher Apex Purification Systems
-
-
-
Clinical Chemistry & Hematology
Roche Cobas® C 311 analyzer
Siemens Advia 2120i
-
Cell-based assay
BD LSRFortessa X-20 flow cytometer
BD FACSVerse flow cytometer
-
Automation & Other lab equipment
Tecan Freedom EVO 200 & Tecan Fluent 1800TomTec , Eppendorf epMotion Liquid Handlers
HP D300e Digital Dispenser
Blue Wash
MultidropTM Combi
BioTek405-plate washer
Omni bead ruptor (different size and model)
Spex Sample prep miniG1600
-

-
Full AAALAC Accreditation
-
OLAW Assured
-
History of successful U.S. FDA inspections
-
State of the Art
Technology

In Vitro ADME Case Studies
-
Diazepam in Human Hepatocytes in HEPATOPAC® Format
-
Metabolic Stability Evaluation of Low Clearance Compounds
-
CYP inhibition evaluation of siRNA and peptide drugs in human hepatocytes
-
Metabolic Stability and Metabolite Identification of Diazepam in Human Hepatocytes in HEPATOPAC® Format
HEPATOPAC® is a long-term, micropatterned co-cultured system of hepatocytes and supportive stromal cells. It maintains stable hepatic function for several weeks and is well suited for slowly metabolized compounds. Diazepam is generally considered as a slowly metabolized compound in hepatocyte-based system. The stability of Diazepam as well as metabolite profile were evaluated in human HEPATOPAC® format.
-
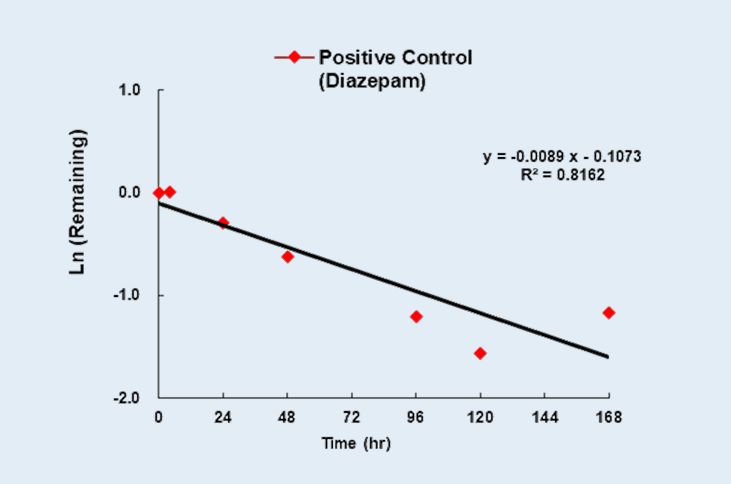
Percent remaining of Diazepam at 1µM in human HEPATOPAC®format as a function of incubation time
Figure 1
-


Metabolic pathway of Diazepam in human HEPATOPAC®format
Figure 2
-
-
Metabolic Stability Evaluation of Low Clearance Compounds using HµREL® Co-culture Human Hepatocytes
The Hurel® co-culture system is a long-term human hepatocytes platform that maintains hepatocyte viability as well as phase I/II enzymes’ activities. This sustained functionality makes it well suited for evaluating test articles with low to moderate intrinsic clearance over extended incubation periods. The metabolic stability of two compounds, ketoprofen (which is primarily metabolized by UGT glucuronidation), and prednisolone (which is primarily metabolized by CYP3A4), were assessed using HµREL® Co-culture Human Hepatocytes system.
-

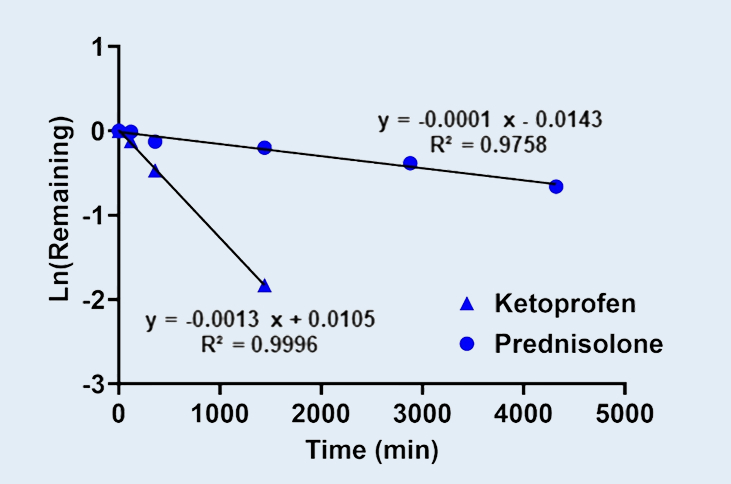
Metabolic Stability of Ketoprofen and Prednisolone in HµREL® Co-culture Human Hepatocytes.
Figure 1
-
-
Cytochrome P450 (CYP) Enzyme Inhibition Evaluation of Inclisiran (siRNA) and Motixafortide (peptide) Using Human Hepatocytes
Human hepatocytes (HH) are preferred over human liver microsomes (HLM) for assessing CYP inhibition of high–molecular‑weight test articles because HH maintain physiologically relevant intracellular exposure, reducing the likelihood of artifactual inhibition that can occur in microsomal systems.
Compared with human liver microsomes (HLM), human hepatocytes (HH) yield fewer false‑positive CYP inhibition results for high–molecular‑weight compounds. Limited membrane permeability in microsomes can lead to disproportionately high enzyme exposure, artificially inflating observed inhibition.
Two high-molecular weight test articles, Inclisiran, an FDA-approved GalNAc-conjugated siRNA and Motixafortide, an FDA-approved synthetic peptide drug, were evaluated using human hepatocytes and human liver microsomes to compare its CYP inhibition potency. As shown in Table 1. Inclisiran did not inhibit CYP3A activity in either human hepatocytes (HH) or human liver microsomes (HLM) test systems. Motixafortide did not inhibit CYP3A in the HH system; however, in the HLM test system, it showed significant inhibition with an IC₅₀ of 5.53 µM. The inhibition observed for Motixafortide in HLM is considered a false positive result.
-

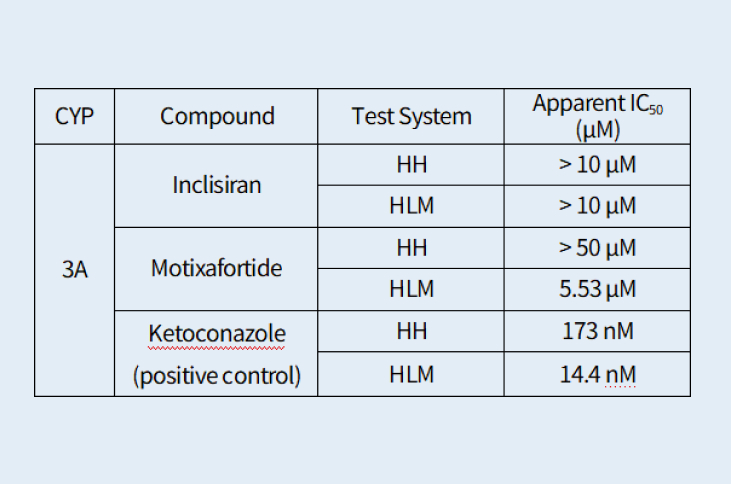
Table 1.
-

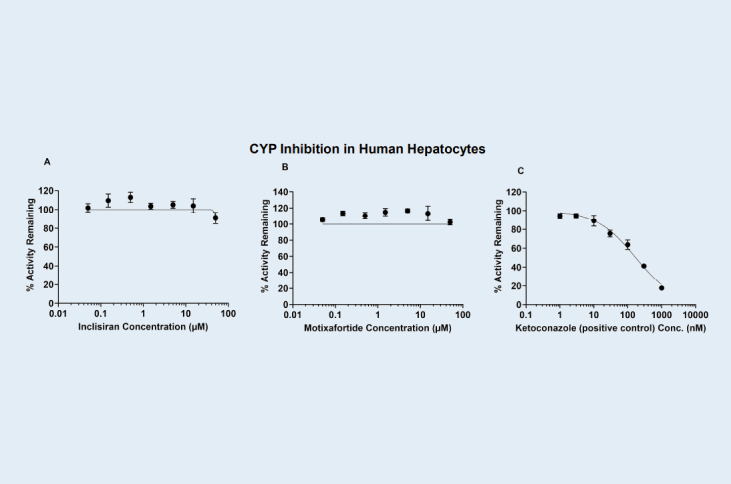
Direct inhibition (DI) of CYP3A activity by Inclisiran, Motixafortide and Ketoconazole (positive control) in human hepatocytes (HH), respectively.
Figure A, B and C.
-

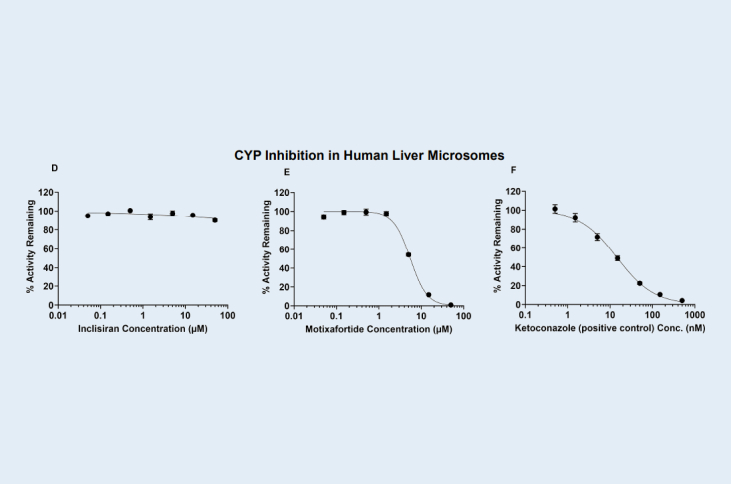
Direct inhibition (DI) of CYP3A activity by Inclisiran, Motixafortide and Ketoconazole (positive control) in human liver microsomes (HLM), respectively.
Figure D, E and F.
In Vivo PK & Radiolabeled ADME Case Studies
-
QWBA of Tissue Distribution
-
Prediction of Human Pharmacokinetics of Therapeutic Monoclonal Antibodies
-
Quantitative Whole Body Autoradiography (QWBA) Characterization of Tissue Distribution Following Administration of a 14C-Labeled Novel Peptide
14C-labeled novel peptide
1-hour intravenous infusion administration: Long-Evans (LE) and Sprague Dawley (SD) rats
LE group time points (starting from the beginning of infusion): 1, 2, 4, 6, 8, 24, 48, 72, 96, 168, 336, 504, 1,344, 2,352, 4,032, and 6,048 hours post-dose
SD group time points (starting from the beginning of infusion): 1, 24, 336, and 504 hours post-dose
Tissues: 55 tissues for analysis; notable tissues showing increased and long-term retention of radioactivity include: aorta, bone, bone marrow, kidney, liver, lung, spleen, and testis
QWBA methodologies conducted
Tissue concentration data via Raytest AIDA Image Analyzer
This study highlights the strength of QWBA methodologies in quantitatively characterizing prolonged tissue retention of a ¹⁴C-labeled peptide following intravenous administration. Persistent radioactivity was detected in renal, skeletal, hematopoietic, and vascular tissues through 6,048 hours post-dose. These findings reinforce the value of QWBA as a critical tool for assessing biodistribution and long-term tissue persistence of emerging large-molecule therapeutics.
-

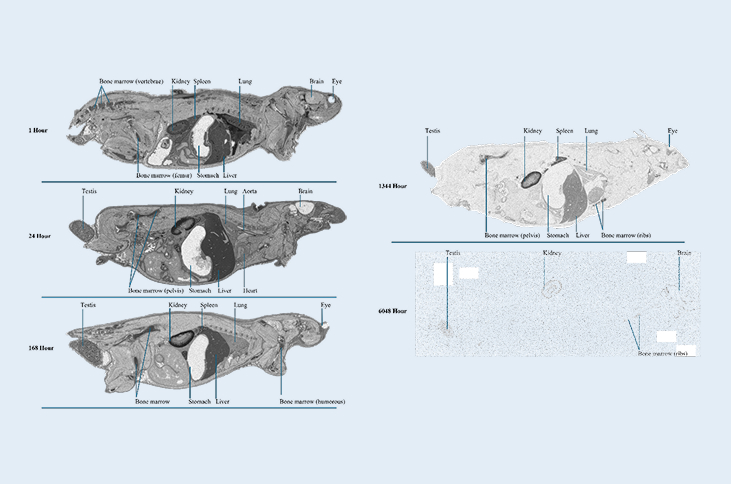
Select autoradioluminogram images (1, 24, 168, 1344, and 6048 hour post-dose) of tissue distribution in the Long-Evans rats
Figure 1.
-
Prediction of Human Pharmacokinetics of Therapeutic Monoclonal Antibodies Using Human FcRn Transgenic Mice
Human FcRn transgenic mice (Tg32 SCID, JAX #018441) were used to evaluate the pharmacokinetics of therapeutic monoclonal antibodies. Because mouse IgG binds weakly to human FcRn, human IgG (1000 mg/kg) was administered intraperitoneally to mimic FcRn competition observed in humans. Test monoclonal antibodies were administered intravenously to mice with or without IgG pretreatment, with dosing occurring 24 hours after IgG pretreatment. Plasma concentrations of the test articles were measured at multiple time points up to 1488 hours using an in-house developed anti-idiotype electrochemiluminescent ELISA method.
IgG pretreatment resulted in varying effects on clearance and half-life across different monoclonal antibodies, reflecting differences in FcRn-mediated recycling. Incorporation of human IgG competition in the Tg32 model improved the translational relevance of the pharmacokinetic profiles and supported better alignment with observed human pharmacokinetics.
-


Plasma concentrations of IgG (A) and antibody test articles (B, C) following IgG pretreatment and subsequent intravenous (IV) administration of the test articles.
Figure 1.
-
Stay Connected
Keep up with the latest news and insights.
















