-
Overview
-
Assays
-
Study Strategies
-
Case Study
-
Regulatory Guidance
-
Experience
-
Instruments
-
FAQs
-
Related Resources
-
Related Services
Overview
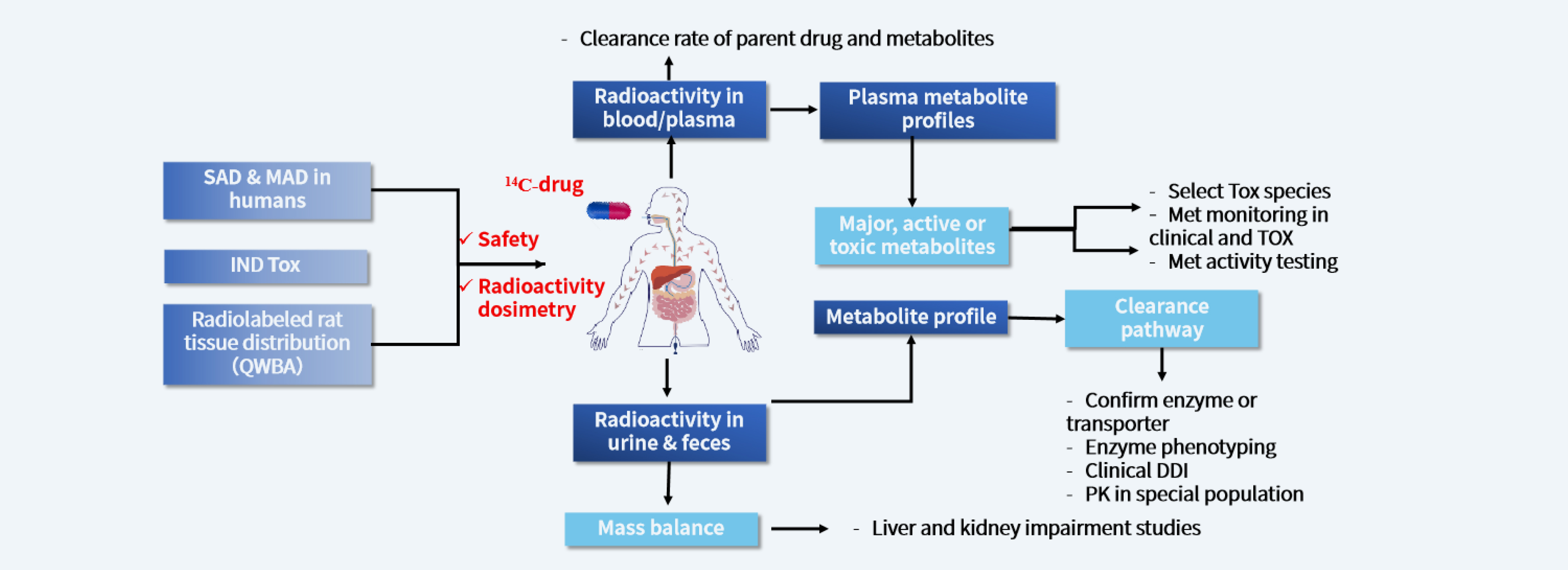
A human radiolabeled (most commonly 14C and 3H) mass balance study is the single most direct method to obtain quantitative and comprehensive information on the absorption, metabolism, and excretion (AME) of the drug in the human body.
The mass balance study can provide information to:
Determine the overall pathways of metabolism and excretion of an investigational drug.
Identify circulating metabolites.
Determine the abundance of metabolites relative to the parent or total drug-related exposure.
The results from mass balance studies help to:
Provide information on which metabolites should be structurally characterized and which metabolites should undergo nonclinical safety assessment or drug-drug interaction (DDI) evaluation.
Assess whether renal or hepatic impairment studies or certain DDI studies are recommended for the investigational drug.
Assess the extent of absorption of the investigational drug with additional data from other studies documenting the investigational drug’s stability in the gastrointestinal tract.
Learn More


Assays
Study Strategies
Case Study
-
Purpose: Compound A is a novel, potent, and highly selective tyrosine kinase inhibitor. This study was conducted to investigate the metabolism, excretion, and pharmacokinetics after a single oral dose to healthy men.
Methods: Six subjects were administrated an oral suspension containing 5 mg of 14C-labeled Compound A (100 μCi) in a fasted state. Blood and excreta samples were collected at the designated time points or intervals for pharmacokinetics and radiometric analyses. Safety assessments were conducted throughout the study.
Results: Over a 336-h post-dose collection period, compound A was well tolerated and absorbed rapidly, mean recovery was 90.11% of the radiolabeled dose, with 60.31% in urine and 29.80% in feces. Parent compound was the predominant circulating component, were cleared from circulation with terminal half-lives more than 30h. M379-3 was the major circulating metabolite, accounting for 17.31% of the total radioactivity. An additional seven circulating metabolites were identified, each accounting for less than 5% of the total radioactivity. In urine, compound A accounted for only 0.50% of the administered dose. three major metabolites M285, M381, and M409-4 were identified accounting for 10.48, 21.16, and 8.92% of the dose, respectively. In feces, compound A accounted for 5.34% of the dose. M205, M365-2, and M380 were the major metabolites, accounting for 2.29, 3.30, and 2.59% of the dose, respectively. Compound A was extensively metabolized prior to excretion, and urine was the major route of excretion.
Learn More
-

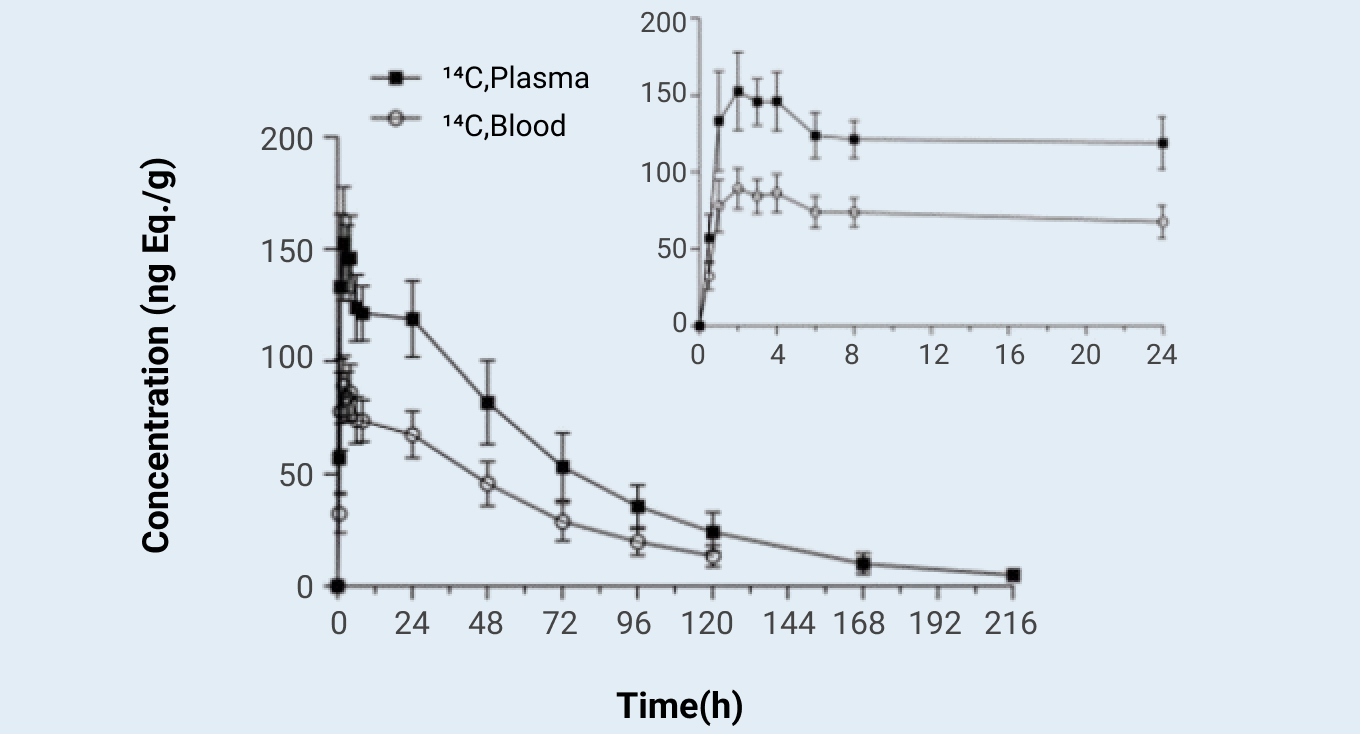
Concentration–time profiles for total radioactivity in blood and plasma after a single oral dose of 5 mg (100 μCi) of [14C] compound A to healthy subjects. Data were mean (SD)
Figure 1
-

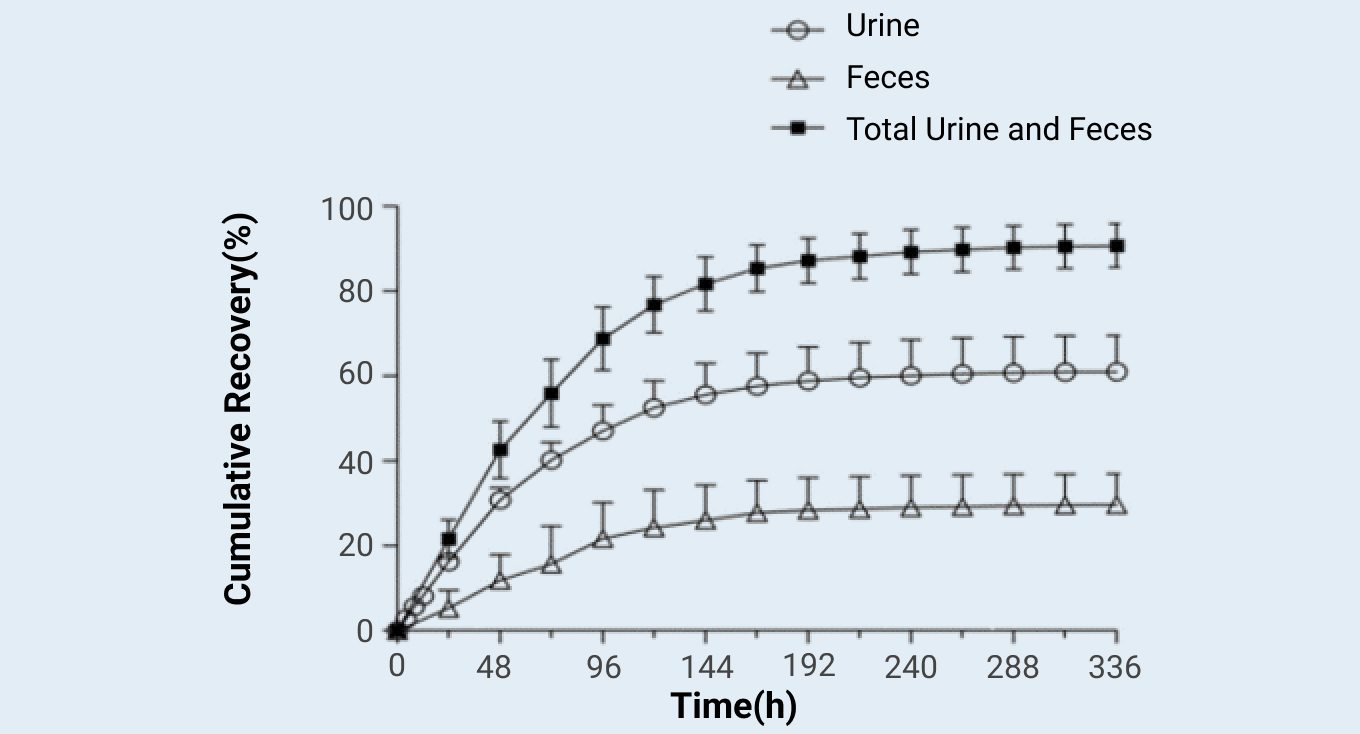
Cumulative excretion of radioactivity in urine and feces from subjects receiving a single oral 5 mg (100μCi) dose of [14C]compound A (N = 6, mean ± SD)
Figure 2
-

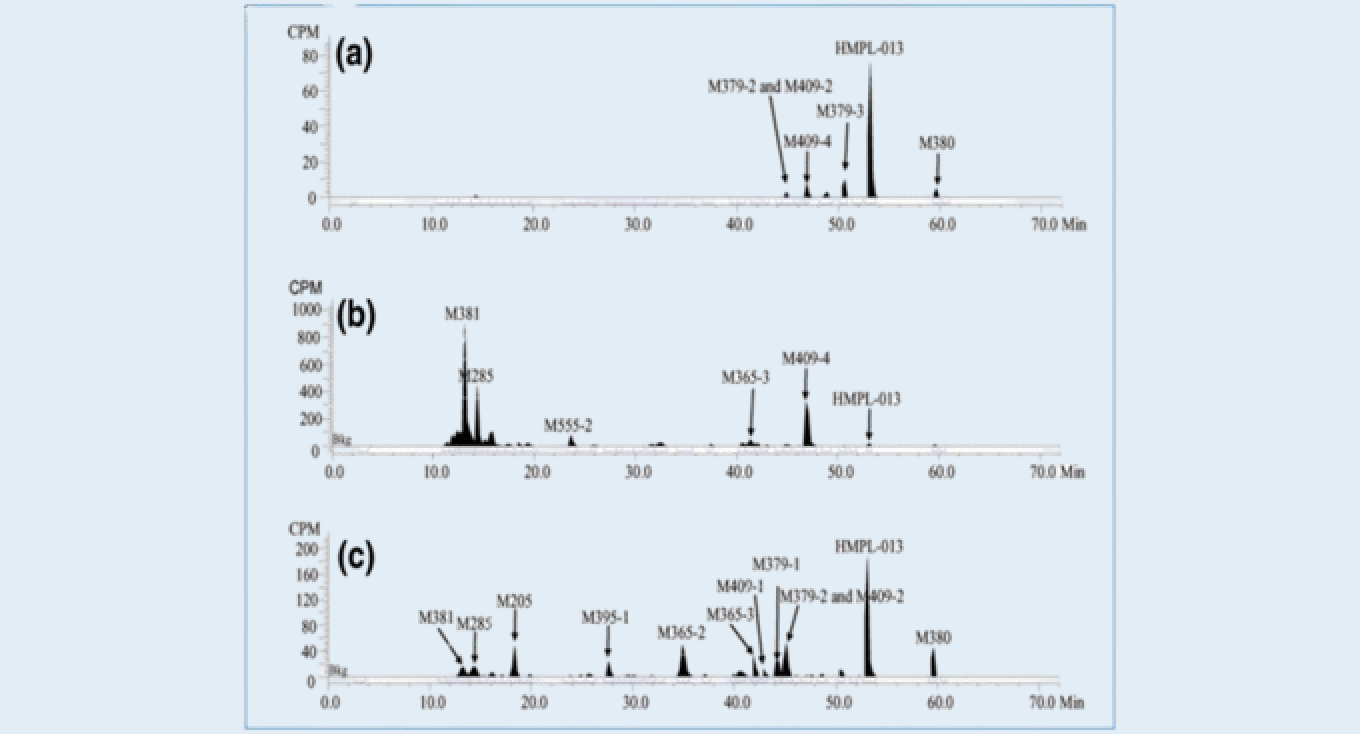
Representative HPLC radio-chromatograms of plasma, urine, and feces1
(a) pooled plasma; (b) pooled urine; (c) pooled fecesFigure 3
-
Experience
-
10+
Years of experience
-
10+
Human mass balance studies each year
-
GCLP system
-
Investigational drugs marketed in NMPA, FDA and EMA
Instruments
-
-


Thermo Q-Exactive™ HF
-


Thermo Q-Exactive™ HF
-


SCIEX API 6500
-


Thermo LTQ-Orbitrap
-


Liquid Scintillation Counter
-


β-RAM Online Radioactivity Detector
-


β-counter Solid Scintillation Counter
-
FAQs
Related Resources




-


GLP-1 Peptide Drug Radiolabeling and ADME Study Strategies
ArticlesDec 24, 2025Learn More -


Decoding Peptide ADME Challenges: Integrated Radiolabeled Synthesis & ADME Platform - DMPK Frontiers
VideosOct 24, 2025Learn More -


Mass balance, Metabolic Disposition, and Pharmacokinetics of [ 14C] Ensartinib, a Novel Potent Anaplastic Lymphoma Kinase (ALK) Inhibitor, in Healthy Subjects Following Oral Administration
PublicationsOct 16, 2025Learn More -


Radiolabeled Synthesis and ADME Services
BrochuresOct 11, 2025Learn More -


The Application of Bile Duct Cannulation Animal Models in Radiolabeled Preclinical ADME Studies
PostersSep 11, 2025Learn More -


Radiolabeling Synthesis and ADME Profiling of GLP-1 Analogs: Strategies and Preclinical Insights
PostersAug 22, 2025Learn More -


Measurement of Phosphoinositide (PIP) Metabolism via Derivatization-Based Liquid Chromatography-Tandem Mass Spectrometry
PostersAug 12, 2025Learn More -


An Integrated Radiolabeling and ADME Platform for Peptide Drugs
WebinarsJul 17, 2025Learn More -


New Book Release: Drug Metabolism and Pharmacokinetics: Frontiers, Strategies, and Applications | WuXi AppTec DMPK
VideosJul 17, 2025Learn More -


Decoding DMPK Frontiers for Novel Therapeutics: WuXi AppTec DMPK’s New Book Release
BlogsJul 10, 2025Learn More -


Drug Metabolism and Pharmacokinetics: Frontiers, Strategies, and Applications
PublicationsJun 13, 2025Learn More -


High-Performance, Professional, Innovative: WuXi AppTec DMPK Accelerates Breakthroughs
VideosMay 22, 2025Learn More -


Metabolic disposition of [14C]-abivertinib, an epidermal growth factor receptor tyrosine kinase inhibitor: Role of glutathione conjugation
PublicationsApr 25, 2025Learn More -


Covalent Drugs DMPK Services
BrochuresApr 10, 2025Learn More -


Absorption, Metabolism and Excretion of Surufatinib in Rats and Humans
PublicationsApr 03, 2025Learn More -


Spotlight on Synthesis of Radiolabeled Compounds with DMPK Considerations and Applications in New Modalities
ArticlesJan 30, 2025Learn More -


WuXi AppTec DMPK: Your Trustworthy Partner
VideosDec 27, 2024Learn More -


Decoding DMPK Profiles of 37 FDA-Approved Peptides: Insights and Radiolabeling Considerations
BlogsOct 18, 2024Learn More -


List of Biological Sample Collection of Quantitative Whole-body Autoradiography (QWBA)
BrochuresAug 21, 2024Learn More -


Metabolite Identification of Nucleoside Analog Prodrugs in Biological Matrix by Derivatization Coupled with Radio-Detector and Mass Spectrometry
PostersMar 07, 2024Learn More -


Radiolabeled Mass Balance Studies in Preclinical Animals
PostersFeb 27, 2024Learn More -


Drug Metabolism and Pharmacokinetics (DMPK) Service
BrochuresOct 27, 2023Learn More -


Quantitative Whole-body Autoradiography (QWBA) Enables ADC Tissue Distribution Studies in Tumor-bearing Nude Rodents
BlogsAug 18, 2023Learn More -


Using Radiolabeling Techniques to Improve ADC Pharmacokinetic Studies
BlogsAug 18, 2023Learn More -


Application of Radiolabeling Techniques in ADC PK Studies
ArticlesJul 26, 2023Learn More -


Quantitative Whole-body Autoradiography (QWBA) vs. Mass Balance Studies
BlogsMay 04, 2023Learn More -


Tissue Distribution Using Quantitative Whole Body Autoradiography (QWBA) of [14c]-Paclitaxel in Tumor Bearing Mice
PostersMay 04, 2023Learn More
Reference
- 1.
Zhou, S., Shao, F., Xu, Z. et al. A phase I study to investigate the metabolism, excretion, and pharmacokinetics of [14C] fruquintinib, a novel oral selective VEGFR inhibitor, in healthy Chinese male volunteers. Cancer Chemother Pharmacol 80, 563–573 (2017). https://doi.org/10.1007/s00280-017-3394-6
Stay Connected
Keep up with the latest news and insights.