-
Overview
-
Services
-
Featured Strengths
-
FAQs
-
Related Resources
-
Related Services
Overview
Radiolabeled ADME studies span all stages of drug development. In screening period, 14C- and 3H- labeled compounds can be used to reduce the difficulty of sample treatment in specific test systems and eliminate the effect of low recoveries. It is recommended to use radiolabeled compounds for endogenous substances to avoid endogenous interference and obtain accurate exogenous data. At PCC stage, the main cause of the low bioavailability can be clearly determined by comparing the post-labeled results (parent and metabolites) and pre-labeled (only parent) results by radioisotope labeling in vivo. At IND stage, radiolabeled compounds can solve the low recovery issue in the mass balance studies. The Quantitative Whole-Body Autoradiography study (QWBA) can obtain complete and detailed tissue distribution information by scanning the animals’ whole body. This can be used to predict the drug efficacy and accumulation degree comprehensively to provide clinical medication guidance. Metabolite radio-profiling and identification studies help understand the clearance pathways, find active/reactive metabolites, and discover human disproportionate/unique metabolites to support the evaluation of drug efficacy and safety.
Learn More


Services
-


Learn More
Radiolabeled Synthesis
[14C] & [3H] radiosynthesis
Full certificate of analysis
-


Learn More
Non-Clinical In Vivo ADME
Blood/plasma pharmacokinetic study
Mass balance; biliary excretion
Traditional tissue distribution (dissection method)
Metabolite profiling & identification
-


Learn More
Quantitative Whole-body Autoradiography (QWBA)
Comprehensive & intuitive tissue distribution studies
Human dosimetry calculation
-


Learn More
Radiolabeled Metabolite Identification
In vitro MetID for cross-species comparision
In vivo MetID in animal plasma, tissue or excreta
In vivo MetID in human plasma and excreta
-


Learn More
Clinical In Vivo AME
Human mass balance study
Blood/plasma pharmacokinetic study
Metabolite profiling & identification
Featured Strengths
-


Experienced and Comprehensive
WuXi AppTec DMPK has over ten years and more than 500 IND and NDA submission experience, we provide radiolabel preclinical and clinical analysis studies.
-


Well Controlled Timeline and Cost
Radiolabeled compounds can be synthesized directly in WuXi AppTec DMPK or provided by the clients. We reduce the cost, shorten shipping time, and design reasonable labeling sites based on chemical structure, synthetic process, and early-stage metabolite identification results.
-


Quick Turnaround Time
By collaborating with multiple hospitals, the shortest time to submit mass balance results can be 24 hours. Preliminary data can help determine whether the experimental expectation is achieved, or the clinical volunteers can be out of the group.
FAQs
-
When is the best time to conduct human radiolabeled mass balance study?
It is usually performed between clinical phase I and phase III studies. It is encouraged to proceed at the early stage of clinical drug development, and it is recommended to carry out at phase IIa, so as to early detect the presence of problems with low mass balance recovery rate, complex metabolites, as well as the presence of high proportion or unique metabolites in plasma.
-
When is it possible not to perform human radiolabeled mass balance study?
Drugs for which mass balance study results can be obtained from acceptable literature sources or FDA-approved product labeling.
Drugs such as monoclonal antibodies, endogenous substances, and analogs (e.g., peptides, hormones, oligonucleotide therapeutics) with known metabolism and elimination pathways based on basic pharmacology and nonclinical ADME information.
Drugs with the majority of the dose (i.e., greater than or equal to 90 percent) recovered in the urine as the unchanged parent drug with minimum metabolism.
Drugs with no or negligible systemic exposures.
-
What are the key benefits of QWBA?
First, to track how compounds of interest are distributed in the body, even for small tissues; second, to assess melanin associated binding in pigmented rodents (like pigmented Long-Evans rats), and calculate human dosimetry dosage from the tissue distribution data.
-
What are the application scenarios of QWBA technology?
In addition to the general tissue distribution studies, QWBA technology also can be used in placental transfer study, tumor distribution study, etc.
-
What is the criteria of radiolabeled compound purity?
For preclinical studies, the purity is recommended to be >95%, and single impurity<1%.
For clinical studies, the purity is recommended to be >98%, and single impurity<1%.
-
What is the regular SA of 14C-labeled compound?
SA is the abbreviation of special activity. The common unit is mCi/mmol. The maximum achievable specific activity for single 14C-labeled organic compound is 62.4 mCi/ mmol. In fact, the specific activity of the synthesized in 14C-labeled compound is a little lower. It is usually between 40 and 50 mCi/mmol.
Related Resources




-


GLP-1 Peptide Drug Radiolabeling and ADME Study Strategies
ArticlesDec 24, 2025Learn More -


Decoding Peptide ADME Challenges: Integrated Radiolabeled Synthesis & ADME Platform - DMPK Frontiers
VideosOct 24, 2025Learn More -


Mass balance, Metabolic Disposition, and Pharmacokinetics of [ 14C] Ensartinib, a Novel Potent Anaplastic Lymphoma Kinase (ALK) Inhibitor, in Healthy Subjects Following Oral Administration
PublicationsOct 16, 2025Learn More -


Radiolabeled Synthesis and ADME Services
BrochuresOct 11, 2025Learn More -


The Application of Bile Duct Cannulation Animal Models in Radiolabeled Preclinical ADME Studies
PostersSep 11, 2025Learn More -


Radiolabeling Synthesis and ADME Profiling of GLP-1 Analogs: Strategies and Preclinical Insights
PostersAug 22, 2025Learn More -


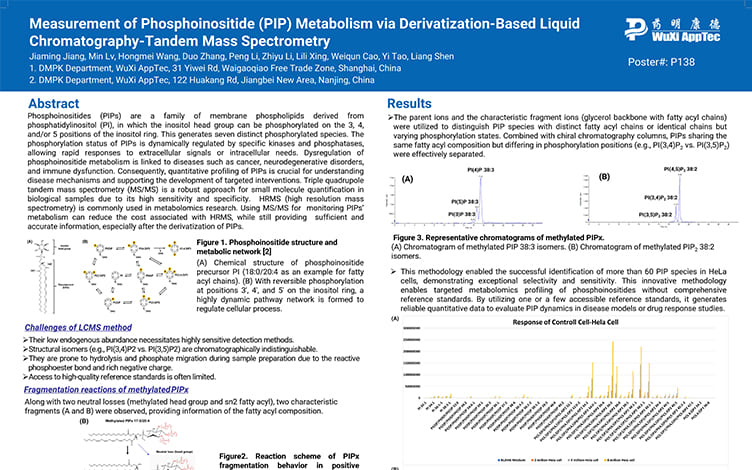
Measurement of Phosphoinositide (PIP) Metabolism via Derivatization-Based Liquid Chromatography-Tandem Mass Spectrometry
PostersAug 12, 2025Learn More -


An Integrated Radiolabeling and ADME Platform for Peptide Drugs
WebinarsJul 17, 2025Learn More -


New Book Release: Drug Metabolism and Pharmacokinetics: Frontiers, Strategies, and Applications | WuXi AppTec DMPK
VideosJul 17, 2025Learn More -


Decoding DMPK Frontiers for Novel Therapeutics: WuXi AppTec DMPK’s New Book Release
BlogsJul 10, 2025Learn More -


Drug Metabolism and Pharmacokinetics: Frontiers, Strategies, and Applications
PublicationsJun 13, 2025Learn More -


High-Performance, Professional, Innovative: WuXi AppTec DMPK Accelerates Breakthroughs
VideosMay 22, 2025Learn More -


Metabolic disposition of [14C]-abivertinib, an epidermal growth factor receptor tyrosine kinase inhibitor: Role of glutathione conjugation
PublicationsApr 25, 2025Learn More -


Covalent Drugs DMPK Services
BrochuresApr 10, 2025Learn More -


Absorption, Metabolism and Excretion of Surufatinib in Rats and Humans
PublicationsApr 03, 2025Learn More -


Spotlight on Synthesis of Radiolabeled Compounds with DMPK Considerations and Applications in New Modalities
ArticlesJan 30, 2025Learn More -


WuXi AppTec DMPK: Your Trustworthy Partner
VideosDec 27, 2024Learn More -


Decoding DMPK Profiles of 37 FDA-Approved Peptides: Insights and Radiolabeling Considerations
BlogsOct 18, 2024Learn More -


List of Biological Sample Collection of Quantitative Whole-body Autoradiography (QWBA)
BrochuresAug 21, 2024Learn More -


Metabolite Identification of Nucleoside Analog Prodrugs in Biological Matrix by Derivatization Coupled with Radio-Detector and Mass Spectrometry
PostersMar 07, 2024Learn More -


Radiolabeled Mass Balance Studies in Preclinical Animals
PostersFeb 27, 2024Learn More -


Drug Metabolism and Pharmacokinetics (DMPK) Service
BrochuresOct 27, 2023Learn More -


Quantitative Whole-body Autoradiography (QWBA) Enables ADC Tissue Distribution Studies in Tumor-bearing Nude Rodents
BlogsAug 18, 2023Learn More -


Using Radiolabeling Techniques to Improve ADC Pharmacokinetic Studies
BlogsAug 18, 2023Learn More -


Application of Radiolabeling Techniques in ADC PK Studies
ArticlesJul 26, 2023Learn More -


Quantitative Whole-body Autoradiography (QWBA) vs. Mass Balance Studies
BlogsMay 04, 2023Learn More -


Tissue Distribution Using Quantitative Whole Body Autoradiography (QWBA) of [14c]-Paclitaxel in Tumor Bearing Mice
PostersMay 04, 2023Learn More
Stay Connected
Keep up with the latest news and insights.