In modern drug discovery, nucleoside analogs have emerged as essential therapeutic agents for treating viral infections and cancer. Because these compounds mimic the fundamental building blocks of life, they require sophisticated analytical techniques to track their transformation within the body. This article explores the mechanisms of nucleosides and their analogs, including nucleotides and nucleoside phosphates, and reviews the latest advances of approved drugs, with a focus on bioanalysis for nucleosides and their phosphates. Overcoming three major analytical hurdles: structural similarity, metabolic complexity, and matrix interference, we systematically summarized the analytical strategy based on LC-MS/MS for nucleosides and their mono-, di-, and triphosphate forms.
What is a Nucleoside?
Nucleosides are compounds formed by a nitrogenous base and a sugar connected by a glycosidic bond. They are classified into ribonucleosides and deoxyribonucleosides, serving as the main components of RNA and DNA. Nucleoside analogs are derivatives obtained through artificial synthesis or chemical modification (of the base, sugar ring, or glycosidic bond) of natural nucleosides. These compounds typically must be phosphorylated by intracellular kinases into their corresponding nucleoside triphosphate analogs to exert their primary therapeutic effects.
As the fundamental units of genetic material, nucleosides have become critical targets in modern drug discovery. They operate within a precise cellular "currency" system, where nucleosides are converted into phosphorylated products such as nucleotides, nucleoside di-/triphosphates, that serve as energy carriers and signaling molecules. Nucleoside drugs, such as Remdesivir, mimic this "cellular currency." Upon entering the cell, they undergo sequential phosphorylation to transform into their active triphosphate forms, precisely targeting viruses or cancer cells.
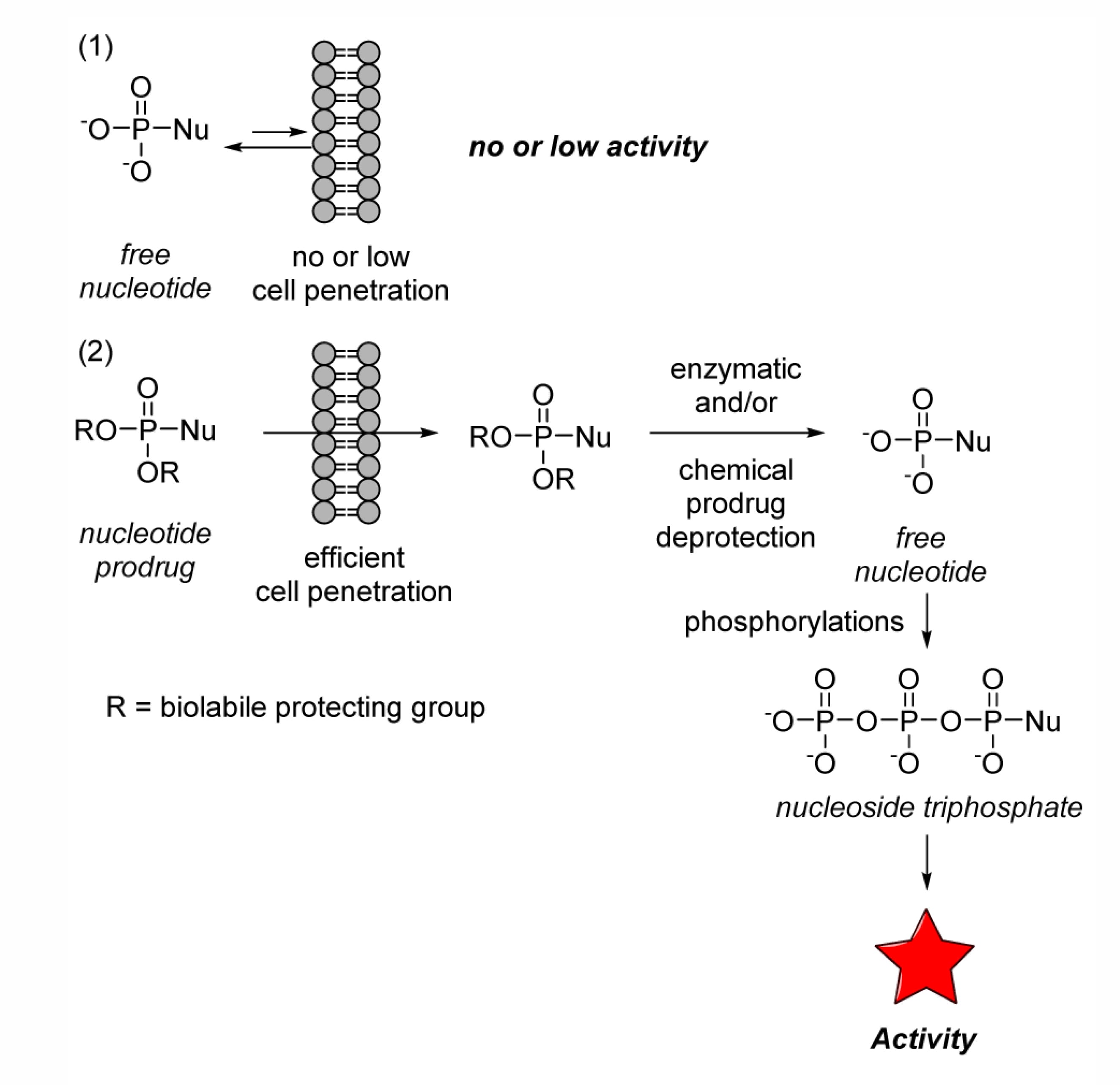
Figure 1. Biological activity regulation mechanism of nucleoside monophosphate prodrugs [1]
Comparison of Nucleosides, Nucleotides, and Phosphates
Nucleosides, nucleotides, and nucleoside phosphates participate in core life processes, including genetic information transfer, energy storage and conversion, and cell signal regulation, through interconversion and synergistic effects in vivo.
Nucleotides
Nucleotides are formed by connecting a purine or pyrimidine base to the 1'-carbon of a pentose (ribose or deoxyribose) via a β-N-glycosidic bond, followed by phosphorylation at the 5'-hydroxyl group. Their biosynthesis involves two major metabolic pathways: de novo synthesis (stepwise assembly of the base ring from small molecule precursors) and salvage synthesis (recycling of free bases or nucleosides).
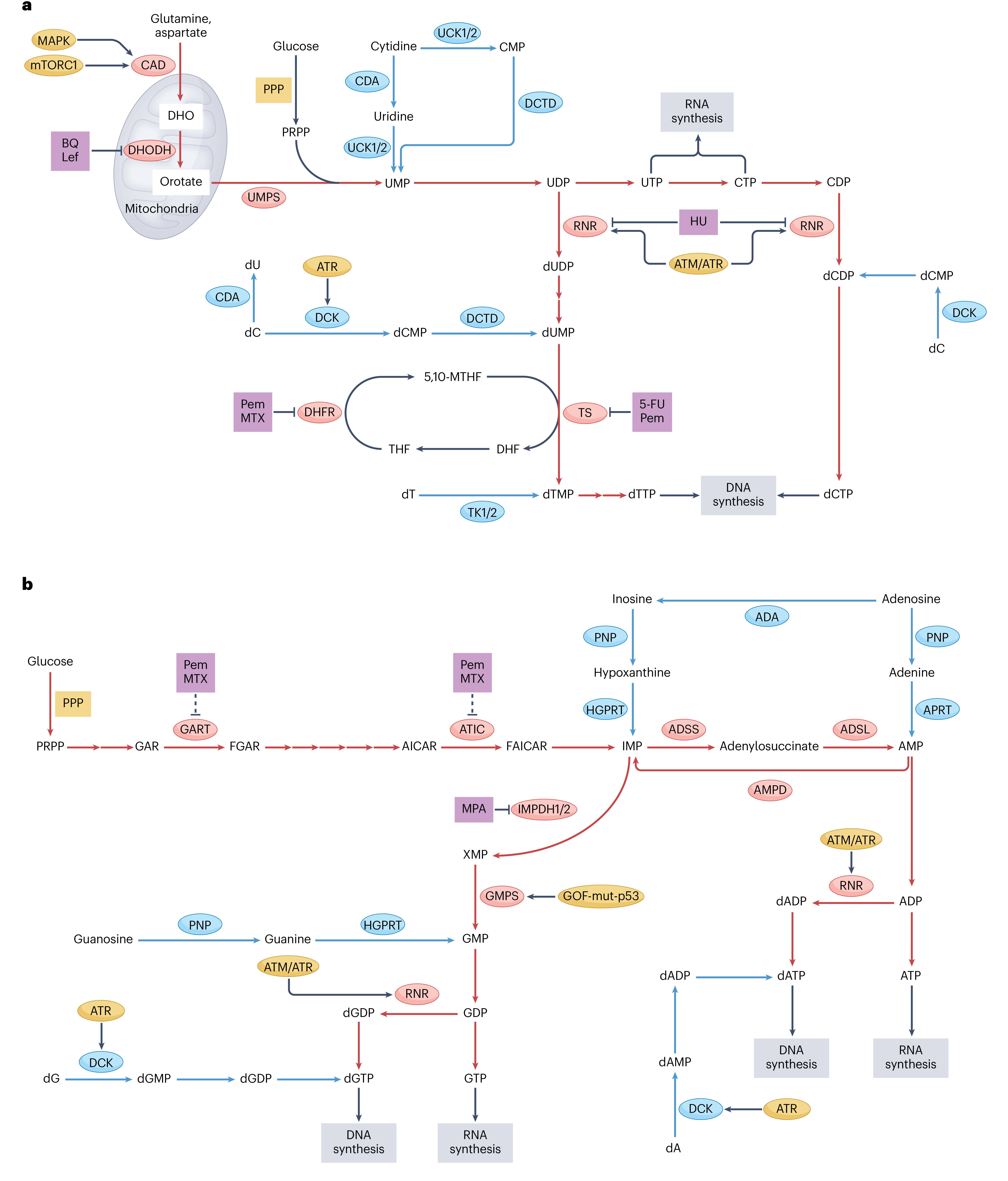
Figure 2. Biosynthetic pathways of pyrimidine and purine nucleotides, related inhibitors, and oncogenic regulators [2]
Nucleotide analogs retain the core structure of natural nucleotides, with at least one phosphate group (P) attached to the sugar moiety. Their chemical diversity is achieved through modifications such as halogenation, introduction of nitrogenous groups (azidation), or masking polarity to improve lipophilicity. These modifications enhance oral absorption and targeted delivery, allowing for the controlled release of the active drug in vivo. They have shown significant efficacy in antiviral therapies (e.g., HCV, HSV) and anti-inflammatory treatments, driving the development of prodrug designs and delivery systems that target nucleic acid metabolism.
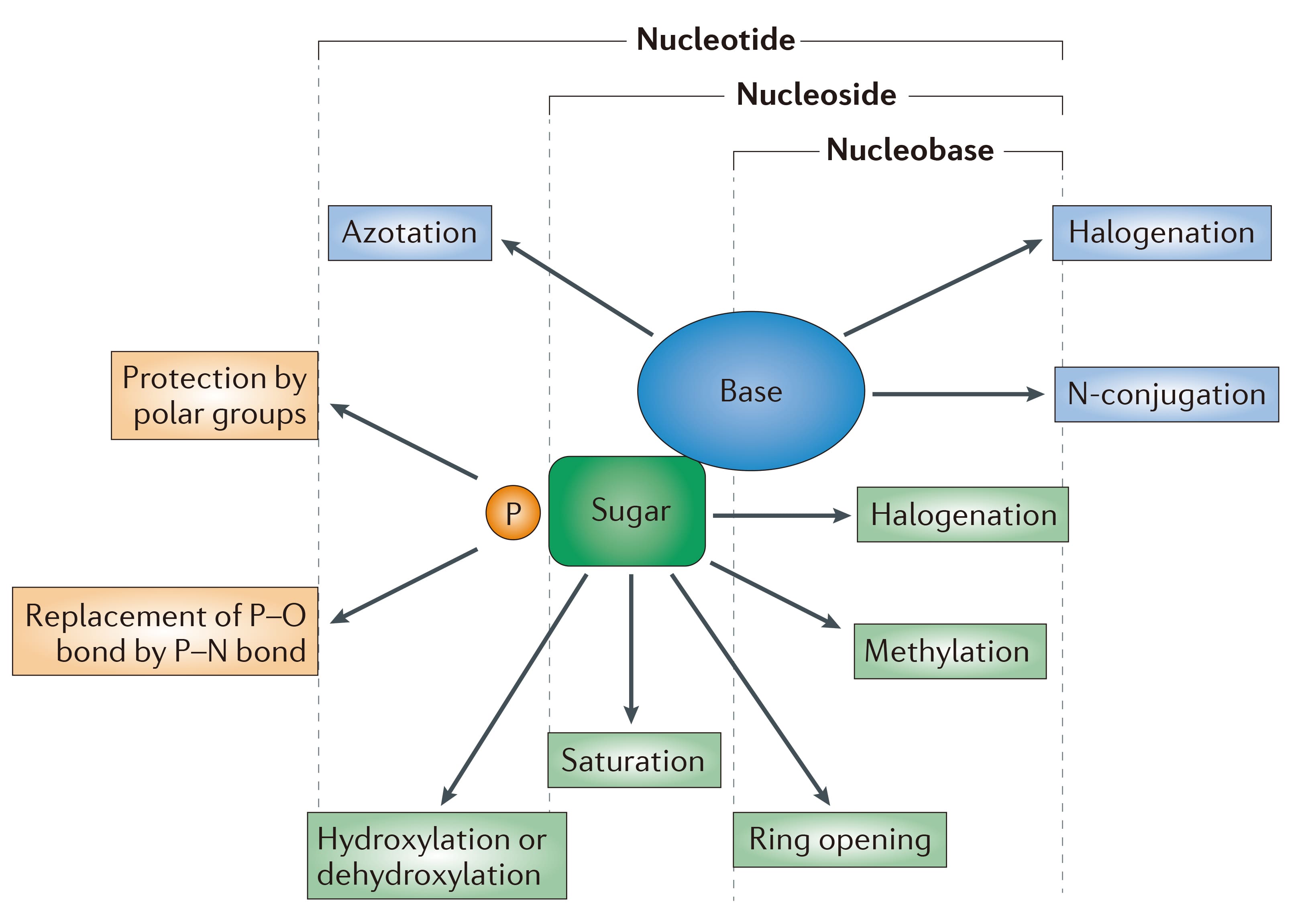
Figure 3. General structure and chemical modifications of nucleotide analogs [3]
Nucleoside Phosphates
Nucleoside phosphate compounds are natural nucleotides chemically modified to possess unique biological activities and pharmacological properties. Their biosynthesis relies on two pathways: synthesis from simple precursors via enzymatic reactions in tissues like the liver, or generation via the kinase-mediated salvage pathway from existing nucleosides or bases.
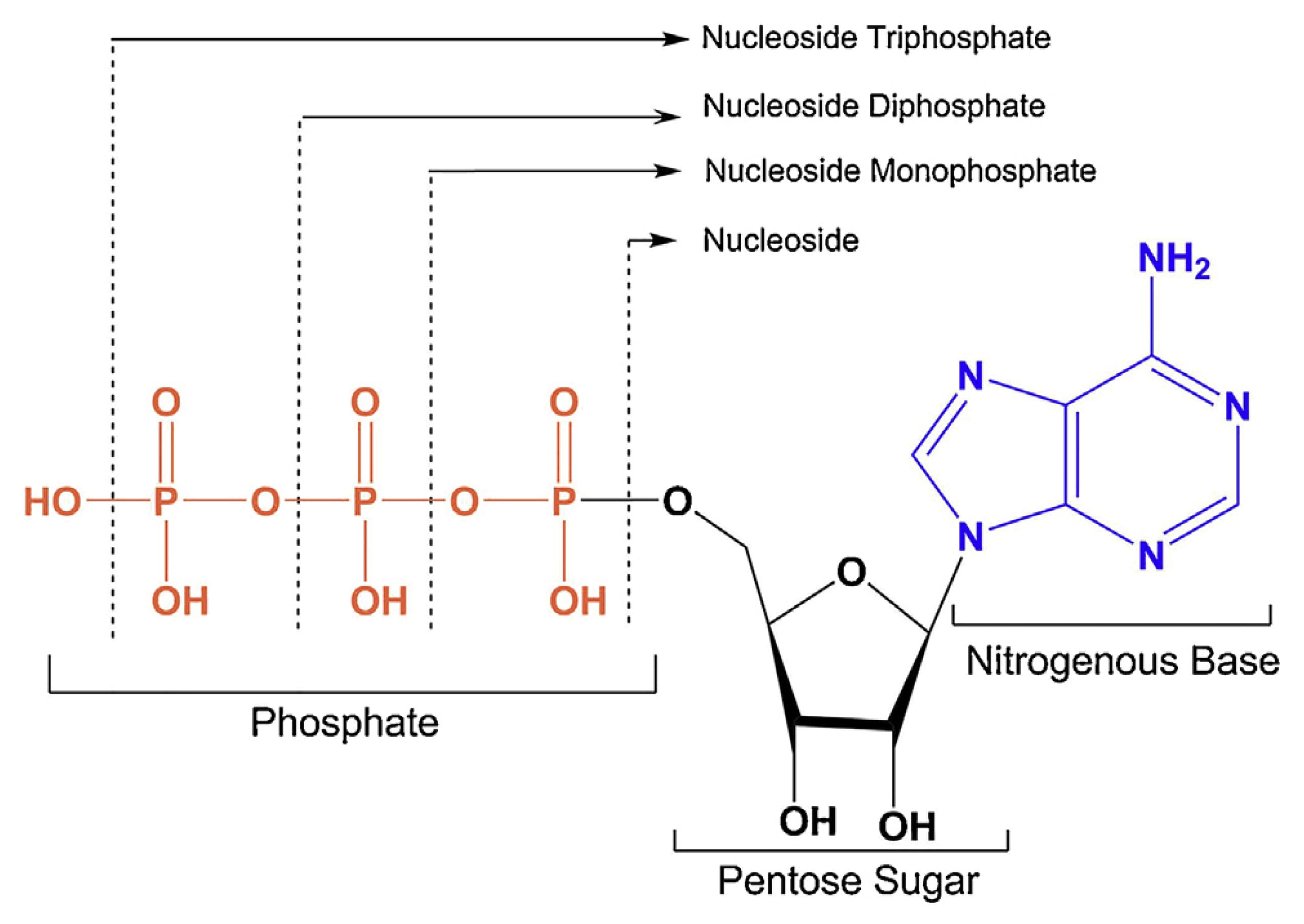
Figure 4. Representative chemical structures of nucleoside triphosphates [4]
Nucleosides, nucleotides, and their analogs exert anti-tumor and antiviral effects by targeting nucleic acid metabolic enzymes. The continuous elucidation of their mechanisms has not only refined drug design but also revealed potential new therapeutic targets.
Therapeutic Landscape and Emerging Trends for Nucleoside Analogs
Nucleoside and nucleotide drugs occupy a critical position in antiviral, anti-tumor, and immunomodulatory fields. Since 1953, targeted nucleotides have been designed as chemotherapy agents to treat cancer, blocking nucleotide metabolism to enhance immune function and achieve therapeutic effects.
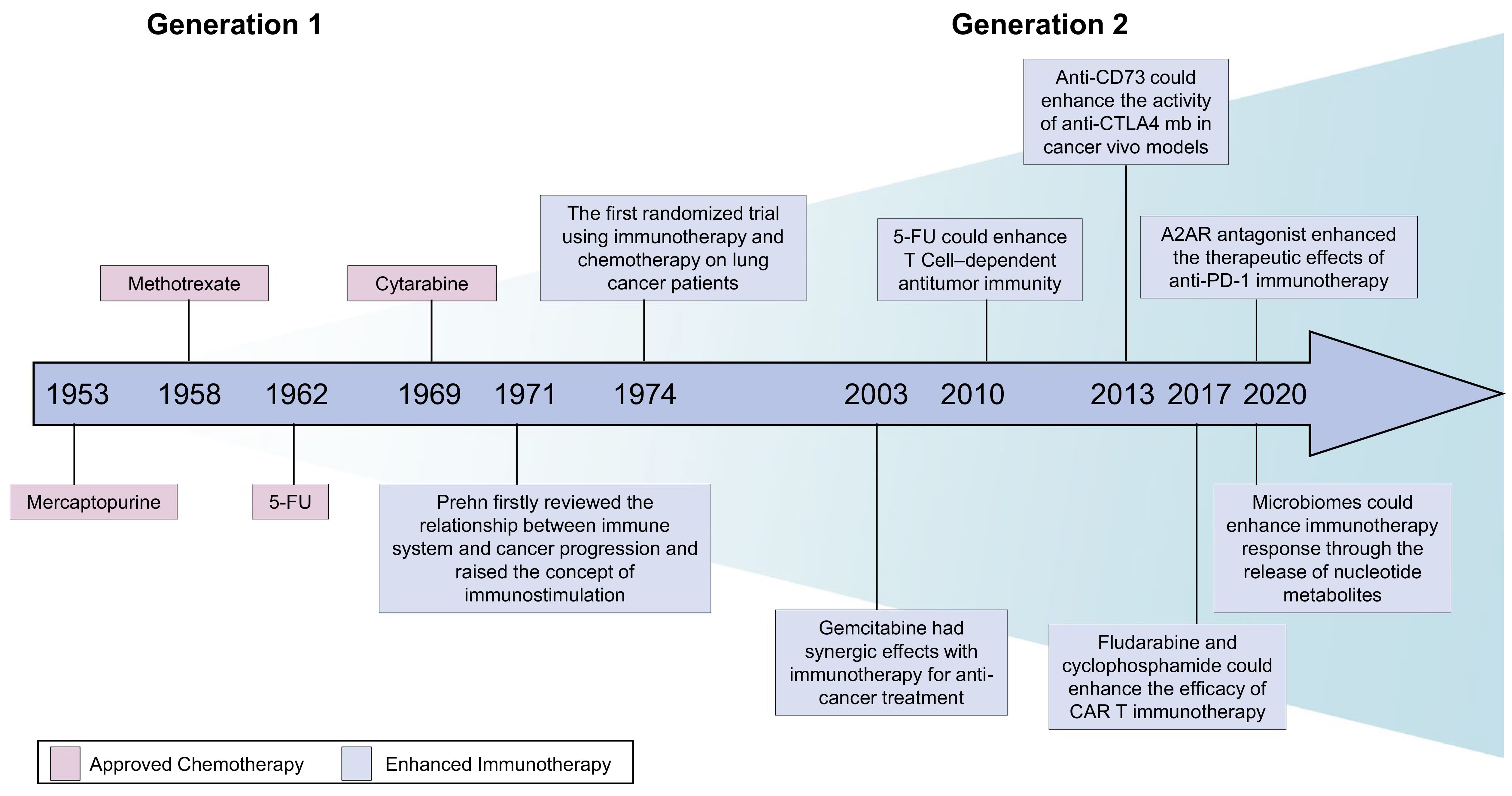
Figure 5. Historical development and breakthroughs in targeting nucleotide metabolism for cancer therapy [5]
Recently, with the rising incidence of major diseases such as viral hepatitis, HIV/AIDS, and cancer, the market demand for nucleoside and nucleoside phosphate drugs continues to expand.
Table 1. Representative approved nucleoside and nucleoside phosphate drugs (domestic and international)
Drug Name | Indication | Developer | Launch Year |
Tenofovir Amibufenamide | Chronic Hepatitis B | Gilead Sciences | 2018 |
Tenofovir Amibufenamide (Domestic) | Chronic Hepatitis B | Hansoh Pharma | 2021 |
Glecaprevir/Pibrentasvir | Pan-genotypic Hepatitis C | AbbVie | 2018 |
Ravidasvir | Hepatitis C | Ascletis Pharma | 2022 |
Dolutegravir/Lamivudine | HIV Infection | GSK | 2019 |
Remdesivir | COVID-19 (Hospitalized) | Gilead Sciences | 2020 |
Nucleoside and analog drugs tend to interact with enzymes involved in DNA or RNA replication, thereby inhibiting the life cycle of viruses or cancer cells. In viral replication, nucleoside drugs effectively block the extension of the viral RNA chain, terminating replication prematurely.
Taking Remdesivir as an example: it is an adenosine analog prodrug with a 1'-cyano modification and a phosphoramidate structure. After hydrolysis by cytoplasmic carboxylesterases, it is sequentially converted into the intermediate GS-441524 monophosphate and finally the active triphosphate metabolite, GS-443902. This triphosphate metabolite mimics ATP to competitively inhibit the viral RdRp (IC50 = 1.1 μM), causing the termination of viral RNA synthesis and exerting anti-SARS-CoV-2 effects [6].
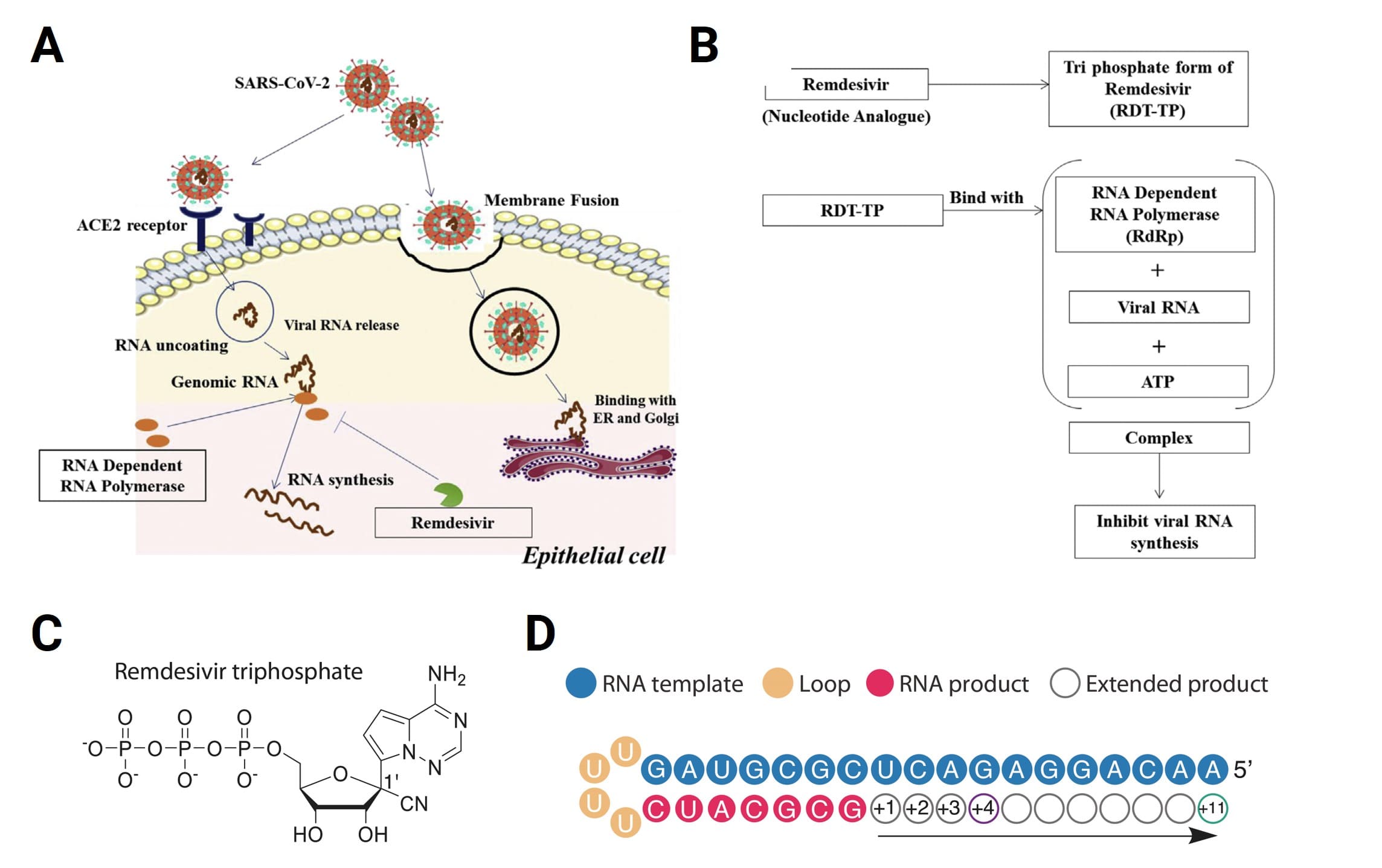
Figure 6. Molecular mechanism of Remdesivir inhibiting SARS-CoV-2 viral replication and hindering RNA extension [7]
Nucleoside phosphates possess multidimensional core functions in other areas:
Oncology: They target and inhibit key enzymes in nucleotide metabolism (e.g., TSase), blocking the metabolic reprogramming pathways required for cancer cell proliferation and metastasis.
Metabolic regulation: The design of 5′-mono/di/triphosphate prodrugs significantly improves drug activation efficiency, while telomerase-mediated nucleoside phosphate metabolism participates in anti-aging mechanisms.
In basic research, the structural analysis of metabolic enzymes like kinases/phosphatases provides a precise molecular basis for targeted drug design. This complete research chain from mechanism exploration to clinical translation has driven breakthroughs in drug development. Simultaneously, mRNA vaccines, functioning as genetic information carriers, have achieved precision similar to epigenetic regulation through base modifications (e.g., modified nucleosides). This technology was awarded the 2023 Nobel Prize for its revolutionary application in disease treatment.
Why LC-MS/MS Analysis Matters for Nucleosides and Their Analogs
Given the multi-target therapeutic potential demonstrated by nucleosides and their phosphorylated products in interfering with the nucleotide metabolic network, establishing precise quantitative analytical methods is a key technical pillar for deciphering their tissue distribution dynamics and optimizing pharmacokinetics-pharmacodynamics (PK/PD).
Bioanalysis is the core method for revealing the in vivo fate of nucleosides and their analogs. However, it faces several distinct challenges:
Isomeric Complexity: Many compounds act like "twins." For example, ATP and dGTP have identical molecular weights and subtle structural differences, making them difficult to separate via liquid chromatography. Analysis requires specific columns and precise pH adjustment of the mobile phase.
Complex Metabolic Pathways: Under kinase action, compounds transform stepwise (Parent Drug → Monophosphate → Diphosphate → Triphosphate). Multi-analyte analysis requires precise quantification and separation of the parent drug and all its phosphorylated forms.
High Polarity and Instability : Phosphate groups are highly polar, making retention on chromatographic columns difficult. Furthermore, polyphosphate forms are prone to enzymatic hydrolysis by phosphatases, necessitating the addition of stabilizers during analysis.
Severe Matrix Interference: Biological samples are often plagued by signal interference from endogenous substances, such as endogenous nucleotides (e.g., cAMP, NAD+). This requires isotope internal standard correction and surrogate matrix quantification.
To address these challenges, Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) has emerged as the gold standard for analyzing nucleoside phosphorylation pathways. Acting as a precise "molecular microscope," it offers:
Superior Separation (HPLC): Distinguishes structurally similar "twin" molecules.
Precise Identification/Quantification (MS/MS, MRM mode): Tracks target molecules with high sensitivity and selectivity.
Dual Capability: Simultaneously confirms identity qualitatively and determines concentration quantitatively.
High-Throughput Potential: Adaptable for large-scale sample analysis.
LC-MS/MS technology not only accurately quantifies parent drugs (like Remdesivir) and their key phosphorylated metabolites but also reveals drug-endogenous nucleoside interactions. This provides critical data support for elucidating efficacy mechanisms and the emergence of drug resistance.
Comprehensive LC-MS/MS Analytical Strategy for Nucleosides and Their Phosphates
Analyzing nucleosides and their phosphate compounds is difficult due to the lack of systematic analysis platforms and strategies. Based on compound characteristics and detection hurdles, we propose targeted analytical methods for parent nucleosides and their converted metabolites (mono/di/triphosphates) by optimizing sample pre-treatment and chromatographic separation.
Parent Nucleoside Drugs
Taking the nucleoside analog prodrug Remdesivir as an example, to overcome its chemical instability, WuXi AppTec DMPK developed a specific stabilizer formula that effectively resolved metabolite degradation issues. Based on LC-MS/MS technology, we achieved the separation and quantitative analysis of the parent drug and its major metabolites.
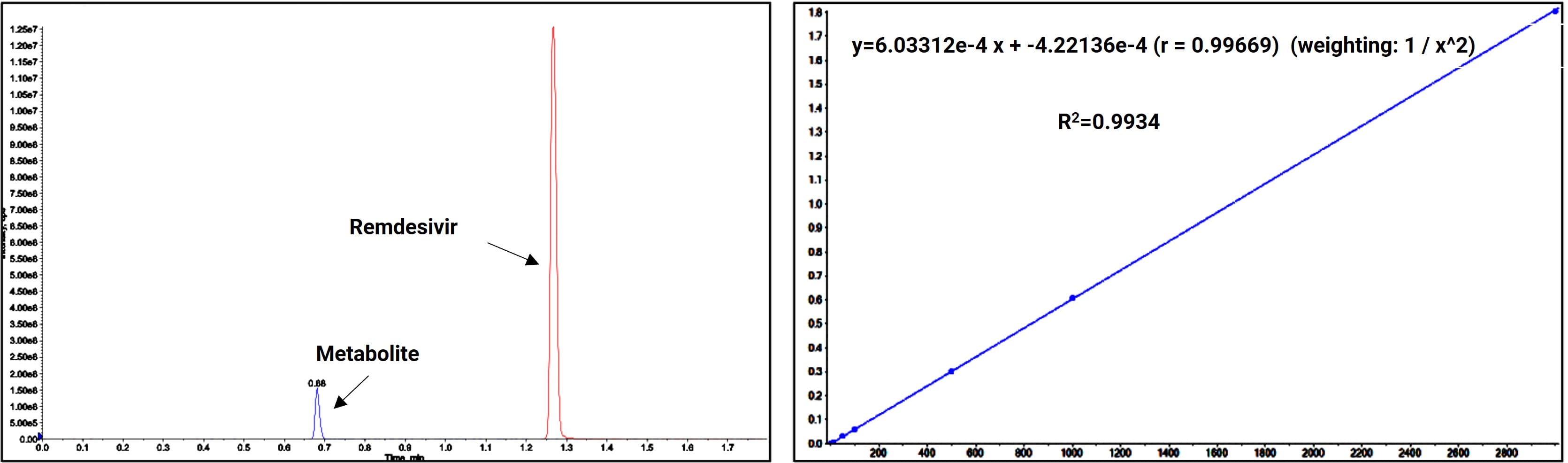
Figure 7. Chromatographic separation and linear regression of Remdesivir metabolites
The established method successfully achieved high-sensitivity and high-selectivity quantitative analysis of the Remdesivir parent drug. It allows for the simultaneous analysis of Remdesivir and its metabolites within two minutes, with a detection limit below 2 ng/mL. However, Remdesivir's antiviral activity relies primarily on its intracellular stepwise phosphorylation into the active triphosphate metabolite. In this critical activation pathway, nucleoside monophosphates and diphosphates are vital intermediates. Analyzing these phosphate forms allows us to better understand the drug's pharmacokinetic behavior and potential efficacy-toxicity correlations.
The following sections focus on analytical strategies for nucleoside mono-, di-, and triphosphates.
Nucleoside Monophosphates (NMPs)
Nucleoside monophosphates are acidic compounds with good water solubility. Because the phosphate group easily forms hydrogen bonds with water molecules, retention on chromatographic columns is difficult. WuXi AppTec DMPK established an NMP analysis method using LC-MS/MS with the following workflow:
Optimization: Refined pre-treatment methods and liquid phase conditions to effectively retain NMPs.
Stabilization: Screened stabilizers to resolve stability issues between NMPs and nucleoside compounds.
Refinement: Adjusted mobile phase pH and gradients, and selected appropriate columns to reduce peak tailing and carryover.
Simplification: Optimized conditions to use common mobile phases (water/acetonitrile system), simplifying the method process.
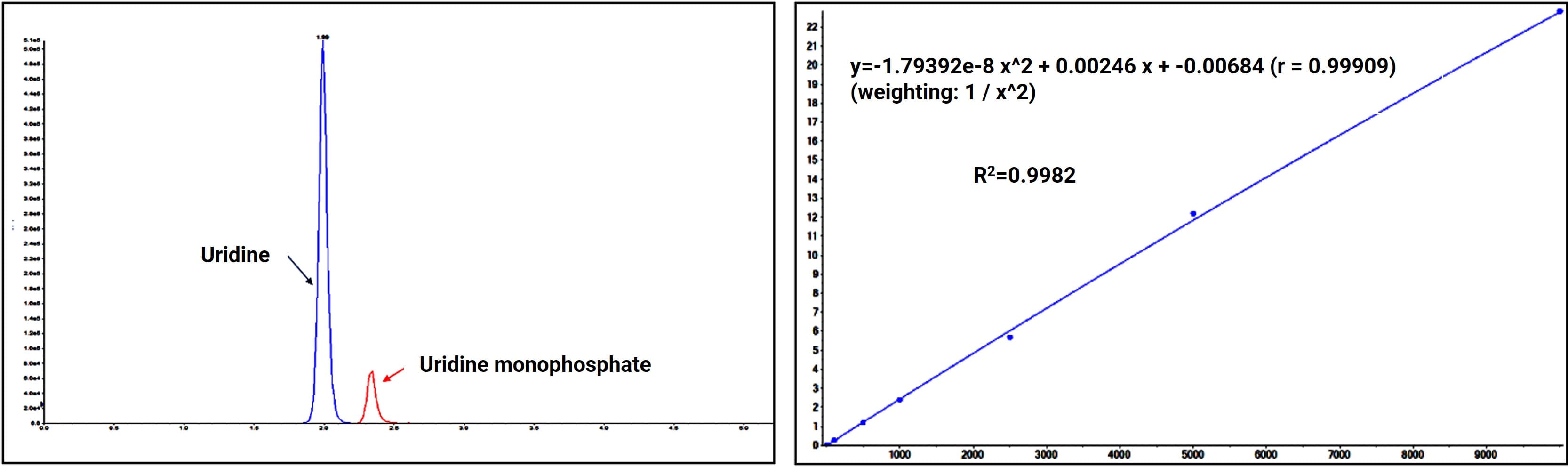
Figure 8. Chromatographic separation and linear regression of uridine and uridine monophosphate
Through continuous exploration, WuXi AppTec DMPK summarized a complete analytical workflow (exemplified by uridine and uridine monophosphate) capable of quickly resolving issues regarding peak shape, retention, and stability, ensuring high-quality and efficient project completion.
Nucleoside Diphosphates (NDPs)
Nucleoside diphosphates contain two phosphate groups, resulting in stronger acidity and increased polarity. They store and release energy via the high-energy bond between the phosphate groups and participate in phospholipid and glycogen synthesis. For example, UDP-glucose is generated via the reaction of glucose-1-phosphate with UTP (uridine triphosphate) and participates in glycogen synthesis in the liver and muscles via glycogen synthase.
WuXi AppTec DMPK broke through the traditional dual-channel ion-pair mobile phase system by developing a single-channel ion-pair LC-MS/MS method. This significantly improved the development efficiency for NDP analysis. Through continuous iteration, reagent consumption was drastically reduced, and column durability was substantially increased. This achieves green, energy-saving operations while significantly lowering the cost per sample.
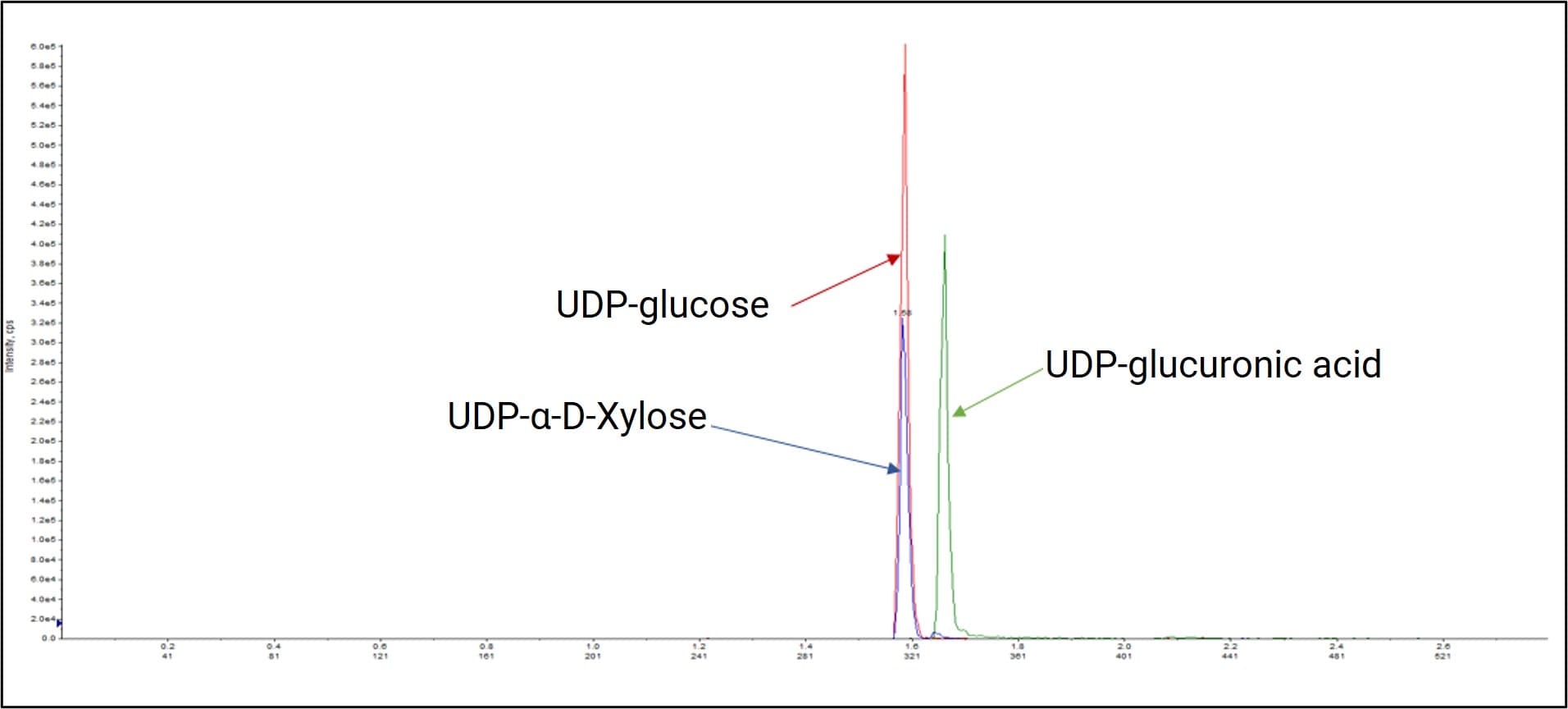
Figure 9. Bioanalysis of UDP-glucose, UDP-α-D-Xylose and UDP-glucuronic acid via LC-MS/MS
Table 2. Comparison of results before and after optimization
Parameter | Basic LC condition | Acidic LC condition |
Amount of ion-pairing reagent | 32.3 g/10,000 injections | 7.32 g/10,000 injections (-77.3%) |
Column lifetime | ~300 injections | ~1000 injections |
Nucleoside Triphosphates (NTPs)
Nucleoside triphosphates possess high-energy phosphate bonds between their three phosphate groups (especially between the β and γ phosphates). Hydrolysis releases massive amounts of energy, driving DNA/RNA synthesis and regulating various physiological functions.
Ribavirin (RBV), used to treat Hepatitis C, exerts antiviral effects through its active phosphorylated metabolite, Ribavirin 5'-triphosphate (RTP). To address the challenge of isobaric interference between endogenous uridine triphosphate (UTP, MW 507.2 Da) and the target RTP, WuXi AppTec DMPK developed a high-selectivity LC-MS/MS method. This successfully achieved baseline separation of the two, validated in red blood cell and liver tissue matrices, effectively eliminating endogenous interference.
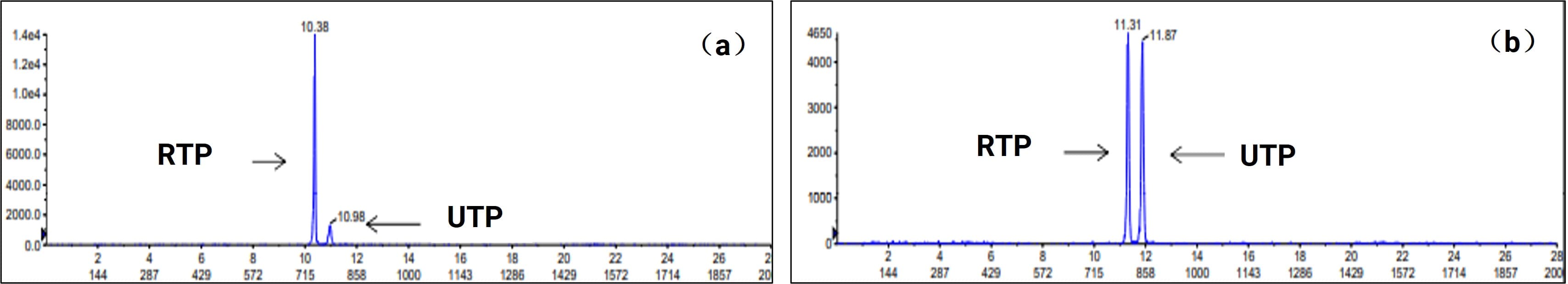
Figure 10. Separation analysis of RTP and UTP via LC-MS/MS in (a) red blood cells and (b) liver tissue
Addressing the separation difficulties caused by the dynamic transformation of NTPs in target organs like the liver, the LC-MS/MS method developed by WuXi AppTec DMPK achieved several key breakthroughs:
High-Throughput Simultaneous Detection: Single-injection quantification of 6 types of NTP/dNTP (12 minutes/sample).
Precise Isomer Separation: Optimized column selectivity to achieve baseline separation of the dGTP/ATP isomer pair.
Ultra-High Sensitivity: Improved mass spectrometry ionization efficiency, reaching an LLOQ of 10 ng/mL in matrix.
Elimination of Phosphate Tailing: Optimized mobile phase pH gradient to obtain symmetrical peak shapes.
Resolution of Endogenous Interference: Rapid screening strategy for surrogate matrices.
Full-Process Stability Assurance: Screened stabilizers (e.g., sodium fluoride) to ensure stability throughout homogenization, sample pre-treatment, and analysis.
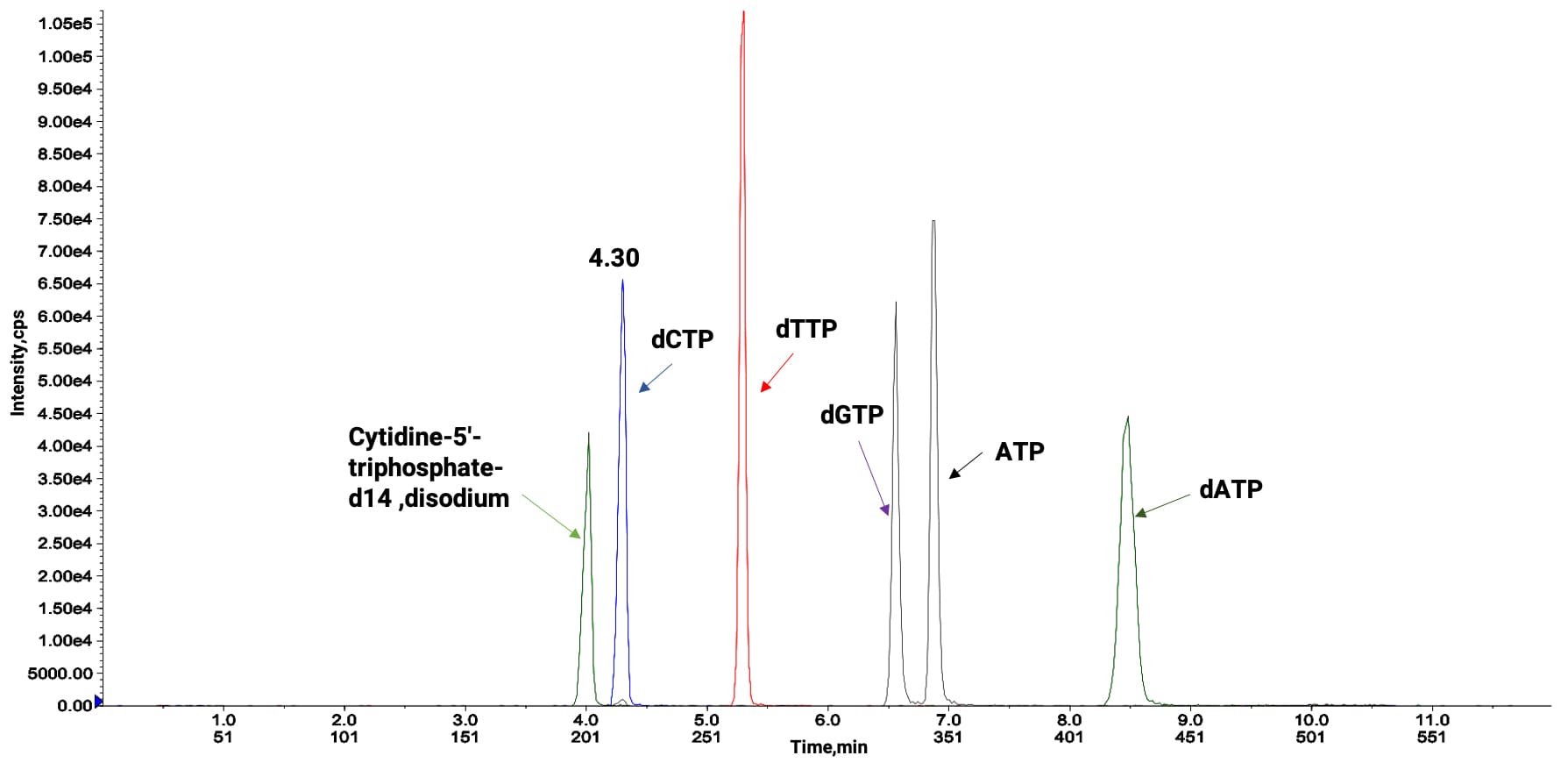
Figure 11. Separation analysis of several marker NTPs via LC-MS/MS
The Bottom Line
WuXi AppTec DMPK LC-MS/MS nucleoside phosphate analysis platform delivers more than just precise quantification; it provides deep analytical insights:
Tracking Activation: Real-time monitoring of how prodrugs (like Remdesivir) are stepwise phosphorylated into their active triphosphate forms within hepatocytes.
Global Impact Insight: Combined with metabolomics, it analyzes changes in the endogenous nucleotide metabolic network (energy metabolism, nucleic acid synthesis) under drug intervention, revealing potential mechanisms of action or off-target effects.
Chiral Separation: Utilizes chiral columns to precisely separate nucleoside enantiomers.
Multidimensional Data Support: Provides a comprehensive, reliable data foundation for efficacy evaluation, pharmacokinetics (PK), pharmacodynamics (PD), and regulatory filings.
WuXi AppTec DMPK has built a mature LC-MS/MS analysis system covering the full "Nucleoside → Monophosphate → Diphosphate → Triphosphate" chain, successfully resolving core challenges regarding stability, retention, separation, interference, and quantification. We are dedicated to providing global partners with high-quality, efficient bioanalytical data, accelerating the progression of nucleoside drugs from target discovery to clinical translation, and jointly advancing the new era of precision medicine.

Figure 12. WuXi AppTec DMPK mass spectrometry analysis platform for nucleosides and their phosphates
Authors: Shuo Zhang, Xianchun Zhang, Wuyun Gong, Zhiyu Li, Lili Xing
Talk to a WuXi AppTec expert today to get the support you need to achieve your drug development goals.
Committed to accelerating drug discovery and development, we offer a full range of discovery screening, preclinical development, clinical drug metabolism, and pharmacokinetic (DMPK) platforms and services. With research facilities in the United States (New Jersey) and China (Shanghai, Suzhou, Nanjing, and Nantong), 1,000+ scientists, and over fifteen years of experience in Investigational New Drug (IND) application, our DMPK team at WuXi AppTec are serving 1,600+ global clients, and have successfully supported 1,700+ IND applications.
Reference
[1] Pradere U, Garnier-Amblard EC, Coats SJ, Amblard F, Schinazi RF. Synthesis of nucleoside phosphate and phosphonate prodrugs. Chem Rev. 2014 Sep 24;114(18):9154-218.
[2] Mullen NJ, Singh PK. Nucleotide metabolism: a pan-cancer metabolic dependency. Nat Rev Cancer. 2023 May;23(5):275-294.
[3] Jordheim LP, Durantel D, Zoulim F, Dumontet C. Advances in the development of nucleoside and nucleotide analogues for cancer and viral diseases. Nat Rev Drug Discov. 2013 Jun;12(6):447-64.
[4] Gautam N, Alamoudi JA, Kumar S, Alnouti Y. Direct and indirect quantification of phosphate metabolites of nucleoside analogs in biological samples. J Pharm Biomed Anal. 2020 Jan 30;178:112902.
[5] Wu HL, Gong Y, Ji P, Xie YF, Jiang YZ, Liu GY. Targeting nucleotide metabolism: a promising approach to enhance cancer immunotherapy. J Hematol Oncol. 2022 Apr 27;15(1):45.
[6] Cox RM, et al. Oral prodrug of remdesivir parent GS-441524 is efficacious against SARS-CoV-2 in ferrets. Nat Commun. 2021 Nov 5;12(1):6415.
[7] Kokic G, et al. Mechanism of SARS-CoV-2 polymerase stalling by remdesivir. Nat Commun. 2021 Jan 12;12(1):279.
Related Services and Platforms




-

 DMPK BioanalysisLearn More
DMPK BioanalysisLearn More -

 Novel Drug Modalities DMPK Enabling PlatformsLearn More
Novel Drug Modalities DMPK Enabling PlatformsLearn More -

 Novel Drug Modalities BioanalysisLearn More
Novel Drug Modalities BioanalysisLearn More -

 Small Molecules BioanalysisLearn More
Small Molecules BioanalysisLearn More -

 Bioanalytical Instrument PlatformLearn More
Bioanalytical Instrument PlatformLearn More -

 PROTAC DMPK ServicesLearn More
PROTAC DMPK ServicesLearn More -

 ADC DMPK ServicesLearn More
ADC DMPK ServicesLearn More -

 Oligo DMPK ServicesLearn More
Oligo DMPK ServicesLearn More -

 PDC DMPK ServicesLearn More
PDC DMPK ServicesLearn More -

 Peptide DMPK ServicesLearn More
Peptide DMPK ServicesLearn More -

 mRNA DMPK ServicesLearn More
mRNA DMPK ServicesLearn More -

 Covalent Drugs DMPK ServicesLearn More
Covalent Drugs DMPK ServicesLearn More
Stay Connected
Keep up with the latest news and insights.