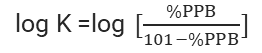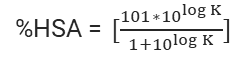Plasma protein binding (PPB) refers to the degree to which a drug binds to proteins within blood plasma. Fraction unbound (fu) is a critical DMPK parameter that needs to be measured accurately, because it has significant impacts on the prediction of drug-drug interactions (DDI), estimations of therapeutic indices (TI), and development of PK/PD relationships[1]. Currently, the primary methods for determining fu in plasma in the early drug discovery stage include equilibrium dialysis (ED), ultracentrifugation (UC), and ultrafiltration (UF). However, conventional techniques frequently encounter difficulties with certain compounds that are highly lipophilic, exhibit poor plasma stability, or are prone to non‑specific binding (NSB). Mechanistic issues—such as adsorption to the dialysis membrane, long incubation time, and degradation during centrifugation—can lead to inadequate analyte recovery, making accurate fu determination challenging[2,3] (as shown in Table 1). Furthermore, for drugs that are highly bound to plasma proteins, analytical methods with extremely high sensitivity are required. This article introduces a novel method for fu determination—human serum albumin (HSA) biomimetic chromatography—and describes its basic principles, experimental method, calibration model, and applications for compounds that are difficult to assess using traditional methods.
Table 1. Common methods for determining PPB
Category | Advantages | Limitations |
Equilibrium Dialysis (ED) |
|
|
Ultrafiltration (UF) |
|
|
Ultracentrifugation (UC) |
|
|
What is HSA Biomimetic Chromatography
Biomimetic chromatography is the name of the High Performance Liquid Chromatography (HPLC) methods that apply stationary phases containing proteins and phospholipids that can mimic the biological environment where drug molecules distribute. Compared to traditional chromatography, biomimetic chromatography more authentically reflects the interactions between drugs and biological components, providing strong support for predicting pharmacokinetic behaviors in vivo [4].
Commonly used biomimetic stationary phases include human serum albumin (HSA), α1-acid glycoprotein (AGP), and immobilized artificial membranes (IAM). All of which have proven valuable in early drug discovery. Over the past two decades, numerous predictive models for volume of distribution, tissue partitioning, protein binding, and blood-brain barrier distribution have been developed using the biomimetic chromatography method. This methodology has been adopted by pharmaceutical companies, including GSK, to investigate hundreds of thousands of compounds[4,5].
HSA is the most abundant plasma protein, accounting for 55% of the total plasma protein content. It serves as the primary carrier for drug binding to plasma proteins and exhibits high binding affinity for acidic, neutral, and some basic drugs. Given its high abundance in plasma and extensive ligand-binding characteristics, this study focuses on HSA biomimetic chromatography [6].
Principles of the HSA Biomimetic Chromatography
HSA biomimetic chromatography employs a stationary phase immobilized with HSA in a high-performance liquid chromatography (HPLC) system to mimic the in vivo protein-binding environment for drugs. When an analyte flows through the column in a mobile phase mimicking physiological conditions (e.g., pH 7.4 buffer), its retention behavior is determined by the binding-dissociation kinetics between the analyte and the immobilized HSA (as shown in Figure 1). Generally, molecules with stronger binding capabilities to HSA have longer retention times on the HSA column, while those with weaker binding have shorter retention times [7]. By establishing a correlation model between the drug's retention time in this biomimetic system and its PPB, fu can be predicted using a simple, rapid chromatographic experiment.
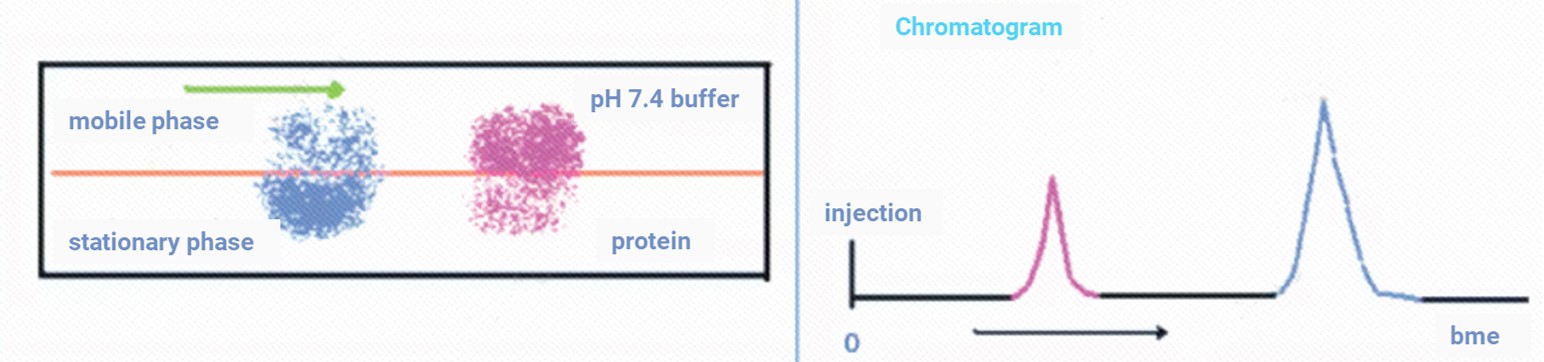
Figure 1. Compound retention depends on interactions between the mobile phase and the stationary phases
Experimental Method of the HSA Biomimetic Chromatography
This method does not require incubation in plasma. The analyte stock solution is diluted to an appropriate concentration using a mixture of ammonium acetate solution (pH 7.4) and isopropanol. After thorough mixing, the sample is analyzed by LC-MS/MS to determine the retention time (tR) of the compound on the biomimetic HSA column.
Table 2. Equations for calculating fu in HSA biomimetic chromatography[8]
Equation.1 |
|
Equation.2 |
|
Equation.3 |
|
Calibration model of the HSA Biomimetic Chromatography
Ten commercially available compounds with human PPB ranging from 5% to 99.5% were selected, covering drugs with low, medium, and high binding. The results demonstrated a strong linear correlation (R2 = 0.9923) between the logarithm of retention time and the linearized %PPB (log K) derived from literature-reported human PPB (as shown in Figure 2).
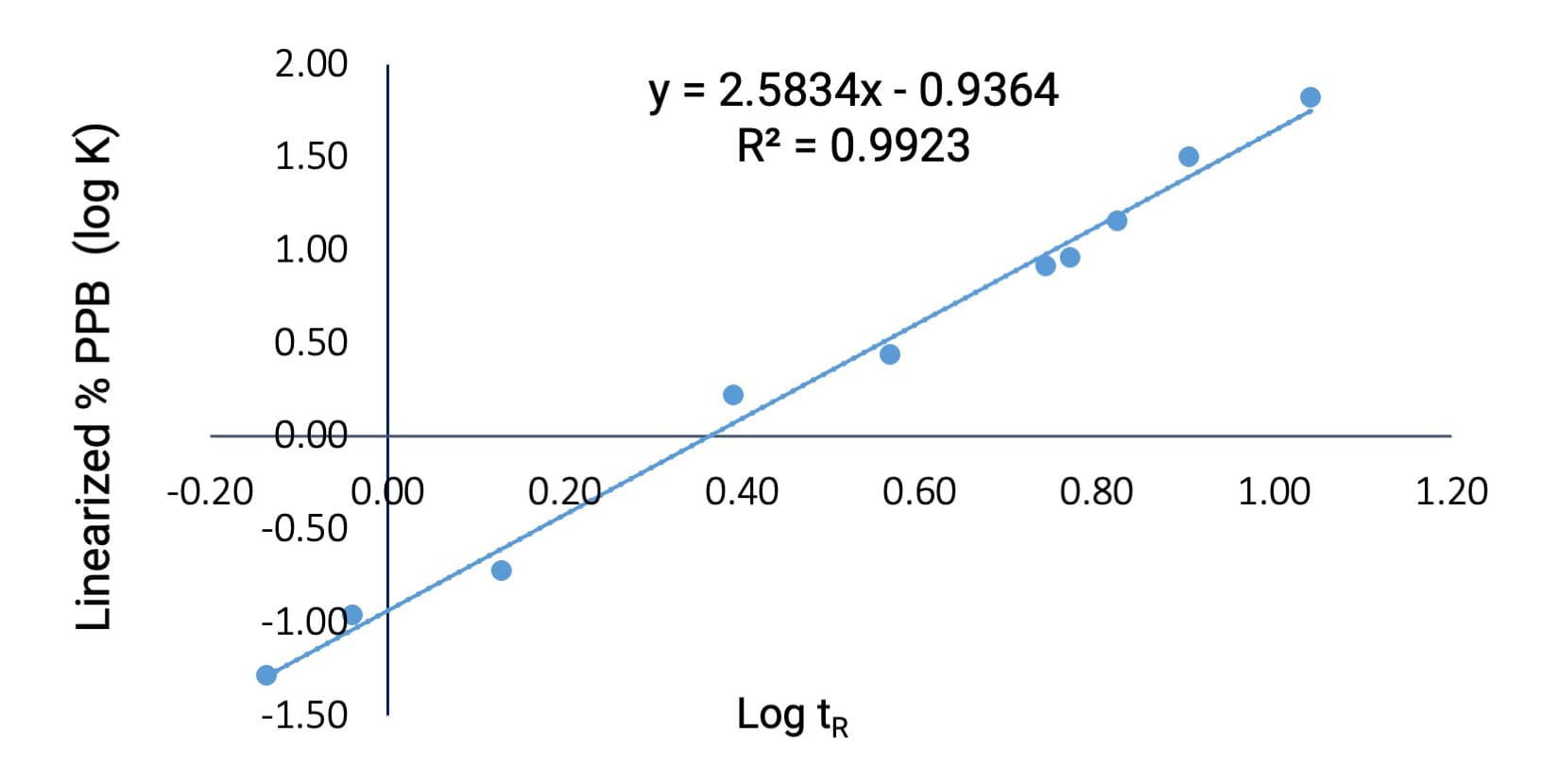
Figure 2. Correlation between retention time and PPB
Key Features of the HSA Biomimetic Chromatography
Compared to equilibrium dialysis, ultracentrifugation, and ultrafiltration, HSA biomimetic chromatography offers the following distinct characteristics for assessing drug PPB:
No plasma incubation required: Simplifies the workflow.
Robustness: Not affected by non-specific binding or by stability and solubility issues in the plasma matrix.
Low sensitivity requirement for the analytical method during sample testing, especially for drugs with high protein binding.
It should be noted that the data reported in the literature reflect total PPB, whereas HSA biomimetic chromatography specifically measures the binding of compounds to HSA. Nevertheless, there is an acceptable correlation between the two methods, indicating that this method can be used for compound PPB screening in the early stages of drug discovery. This finding is also supported by results published by Klara Valkó et al.[9]
Applications of the HSA Biomimetic Chromatography
Case 1: Small Molecule Drug — Captopril
Captopril is an oral ACE inhibitor used to treat hypertension and chronic heart failure. The thiol group in its molecule is prone to oxidation and can form disulfide bonds with itself or endogenous thiol-containing compounds in plasma (such as cysteine and glutathione), resulting in poor stability. This often leads to low recovery and inaccurate fu measurements in traditional PPB assays, which typically require optimized reduction systems (such as TCEP) for reliable results. In contrast, the biomimetic chromatography method does not require plasma incubation and effectively avoids such stability issues. Validation confirmed that the fu value obtained by this method is close to that obtained by ultrafiltration in human plasma (as shown in Table 3).
Table 3. Comparison of fu determination methods for captopril
Compound | Fraction Unbound (fu) | |
Ultrafiltration Method | HSA Biomimetic Chromatography | |
Captopril | 0.8310 | 0.9365 |
Case 2: Peptide — Pasireotide
Pasireotide, an orphan drug used for Cushing's disease and acromegaly, is a 14-amino acid cyclic peptide that is prone to non-specific adsorption, leading to low extraction recovery during sample preparation. Additionally, it exhibits high PPB, making quantification of fu challenging. When biomimetic chromatography was used for fu determination, the value was highly consistent with those obtained by ultracentrifugation in human plasma (as shown in Table 4). This approach effectively minimizes errors caused by adsorption loss and insufficient sensitivity, thereby ensuring the accuracy of the result.
Table 4. Comparison of fu determination methods for pasireotide
Compound | Fraction Unbound (fu) | |
Ultracentrifugation Method | HSA Biomimetic Chromatography | |
Pasireotide | 0.0307 | 0.0294 |
Conclusion
HSA biomimetic chromatography method for evaluating PPB has been developed and validated in our laboratory for nearly 100 compounds, including both small-molecule drugs and peptides. This method is effective for challenging cases with poor plasma stability or strong non-specific binding, making it a valuable complementary strategy for fu determination.
WuXi AppTec DMPK possesses a comprehensive technical system for plasma protein binding (PPB) determination, covering equilibrium dialysis (including HTD and RED), ultracentrifugation, ultrafiltration, flux dialysis, competitive dialysis, biomimetic chromatography, and reporter enzyme methods for dynamic protein binding studies. We can conduct protein binding experiments in various biological matrices such as plasma, tissue homogenates, hepatocytes, and liver microsomes. Furthermore, we support testing for compounds with diverse, complex properties, including unstable compounds, highly bound compounds, covalent inhibitors, peptides, PROTACs, siRNAs, ASOs, etc., providing multi-faceted and highly flexible support for drug development.
Authors: Minxia Li, Chunhong Lu, Mengjie Sun, Xinxin Wen, Jie Wang, Xiaotong Li, Lili Xing
Talk to a WuXi AppTec expert today to get the support you need to achieve your drug development goals.
Committed to accelerating drug discovery and development, we offer a full range of discovery screening, preclinical development, clinical drug metabolism, and pharmacokinetic (DMPK) platforms and services. With research facilities in the United States (New Jersey) and China (Shanghai, Suzhou, Nanjing, and Nantong), 1,000+ scientists, and over fifteen years of experience in Investigational New Drug (IND) application, our DMPK team at WuXi AppTec are serving 1,600+ global clients, and have successfully supported 1,700+ IND applications.
Reference
[1] Di Li. An update on the importance of plasma protein binding in drug discovery and development, Expert Opinion on Drug Discovery, 2021, 16(12): 1453–1465.
[2] Dunja Dimitrijevic, Eric Fabian, Dorothee Funk-Weyer, Robert Landsiedel. Rapid equilibrium dialysis, ultrafiltration, or ultracentrifugation? Evaluation of methods to quantify the unbound fraction of substances in plasma, Biochemical and Biophysical Research Communications, 2023, 651: 114–120.
[3] Brigitte Buscher, Sirpa Laakso, Hermann Mascher, Klaus Pusecker, Mira Doig, Lieve Dillen, Winfried Wagner-Redeker, Thomas Pfeifer, Pascal Delrat, Philip Timmerman. Bioanalysis for plasma protein binding studies in drug discovery and drug development: views and recommendations of the European Bioanalysis Forum, Bioanalysis, 2014, 6(5): 673–682.
[4] Klara Valkó. Biomimetic chromatography—A novel application of the chromatographic principles, Analytical Science Advances, 2022, 3: 146–153.
[5] Shenaz Bunally,Robert J. Young. The role and impact of high throughput biomimetic
measurements in drug discovery. ADMET & DMPK, 2018 6(2): 74-84.
[6] George Lambrinidis, Theodosia Vallianatou, Anna Tsantili-Kakoulidou. In vitro, in silico and integrated strategies for the estimation of plasma protein binding: A review, Journal of Pharmaceutical and Biomedical Analysis, 2015, 86: 27–45.
[7] Klara Valkó. Biomimetic chromatography to accelerate drug discovery: Part 1, LCGC North America, 2018, 36.
[8] Klara Valkó, Shenaz B Nunhuck, Alan P Hill. Estimating unbound volume of distribution and tissue binding by in vitro HPLC-based human serum albumin and immobilised artificial membrane-binding measurements, Journal of Pharmaceutical Sciences, 2011, 100(3): 123–130.
[9] Klara Valkó, Shenaz Nunhuck, Chris Bevan, Michael H. Abraham, Derek P. Reynolds. Fast Gradient HPLC Method to Determine Compounds Binding to Human Serum Albumin. Relationships with Octanol/Water and Immobilized Artificial Membrane Lipophilicity. Journal of Pharmaceutical Sciences, 2003, 92(11): 2236–2248.
Related Services and Platforms




-

 In Vitro ADME ServicesLearn More
In Vitro ADME ServicesLearn More -

 DMPK BioanalysisLearn More
DMPK BioanalysisLearn More -

 Physicochemical Property StudyLearn More
Physicochemical Property StudyLearn More -

 Permeability and Transporter StudyLearn More
Permeability and Transporter StudyLearn More -

 Drug Distribution and Protein Binding StudiesLearn More
Drug Distribution and Protein Binding StudiesLearn More -

 Metabolic Stability StudyLearn More
Metabolic Stability StudyLearn More -

 Drug Interactions StudyLearn More
Drug Interactions StudyLearn More -

 Novel Drug Modalities BioanalysisLearn More
Novel Drug Modalities BioanalysisLearn More -

 Small Molecules BioanalysisLearn More
Small Molecules BioanalysisLearn More -

 Bioanalytical Instrument PlatformLearn More
Bioanalytical Instrument PlatformLearn More
Stay Connected
Keep up with the latest news and insights.