As drug conjugates become a focal point in pharmaceutical R&D, peptide receptor radionuclide therapies (PRRTs) and diagnostic peptide-receptor radionuclide conjugates (PRCs) have entered a pivotal era of development. The 2018 approval of Lutathera (177Lu-Dotatate), the first PRRT, marked the official entry of PRRTs onto the oncology stage. To date, nine PRC drugs have been approved globally, comprising seven diagnostic agents and two therapeutic agents (Novartis’s Lutathera® and Pluvicto®). Radiolabeled octreotide derivatives have been successfully applied in the routine nuclear medicine diagnosis and treatment of neuroendocrine tumors, demonstrating significant clinical translational potential. Furthermore, Bicycle Therapeutics plc recently released its first human imaging data, which not only validated MT1-MMP as a potential new target for cancer therapy but also highlighted the advantages of bicycle radionuclide conjugates (BRCs). This article provides a systematic overview of the structural characteristics and advantages of PRRTs and diagnostic PRCs, their theranostic effect, alongside metabolic properties and considerations for metabolic studies, offering insights to support their development.
Table 1. Approved peptide receptor radionuclide therapies (PRRTs) and diagnostic PRCs
Drug Name | Category | Manufacturer | Target | Indication | Approval Date |
68Ga-PSMA-11 | Diagnostic | Novartis | PSMA | Prostate cancer | Mar 2022 |
Lutetium Lu-177 vipivotide tetraxetan | Therapeutic | Novartis | PSMA | Castration-resistant prostate cancer | Mar 2022 |
TLX591-CDx | Diagnostic | Telix; Grand Pharma | PSMA | Prostate cancer | Dec 2021 |
68Ga-PSMA-11 | Diagnostic | Univ. of California | PSMA | Prostate cancer | Dec 2020 |
Copper Cu-64 dotatate | Diagnostic | RadioMedix | SSTR2 | Neuroendocrine tumors | Sep 2020 |
Gallium Ga-68 edotreotide | Diagnostic | ITM; Grand Pharma | SSTR2 | Neuroendocrine tumors | Aug 2019 |
177Lu-DOTATATE | Therapeutic | Novartis | SSTR2 | GEP-NETs, Neuroendocrine tumors | Jan 2018 |
Gallium Ga-68 edotreotide | Diagnostic | Novartis | SSTR2 | GEP-NETs, Neuroendocrine tumors | Dec 2016 |
Gallium Ga-68 dotatate | Diagnostic | Novartis | SSTR2 | Neuroendocrine tumors | Jun 2016 |
Indium In-111 pentetreotide | Diagnostic | Curium | SSTR2 | Neuroendocrine tumors | Jun 1994 |
What are PRRTs and PRCs
The fundamental structure of a PRC typically consists of a radionuclide and a targeting peptide. More complex conjugation systems may include auxiliary units such as chelators and linkers (Figure 1). The primary distinction between PRCs and antibody-drug conjugates (ADCs) is that the payload is not a cytotoxic small molecule, but a radionuclide with diagnostic or therapeutic capabilities. When used for therapy, this modality is known as peptide receptor radionuclide therapy (PRRT). Radionuclides function via emitted radiation and do not necessarily need to enter the cell; similarly, the linker does not require cleavage to release an active payload, thereby enhancing the in vivo safety and stability of the PRC. The peptide moiety provides precise positioning, delivering the radionuclide to the target organ to enable theranostic (integrated diagnosis and therapy) functions.
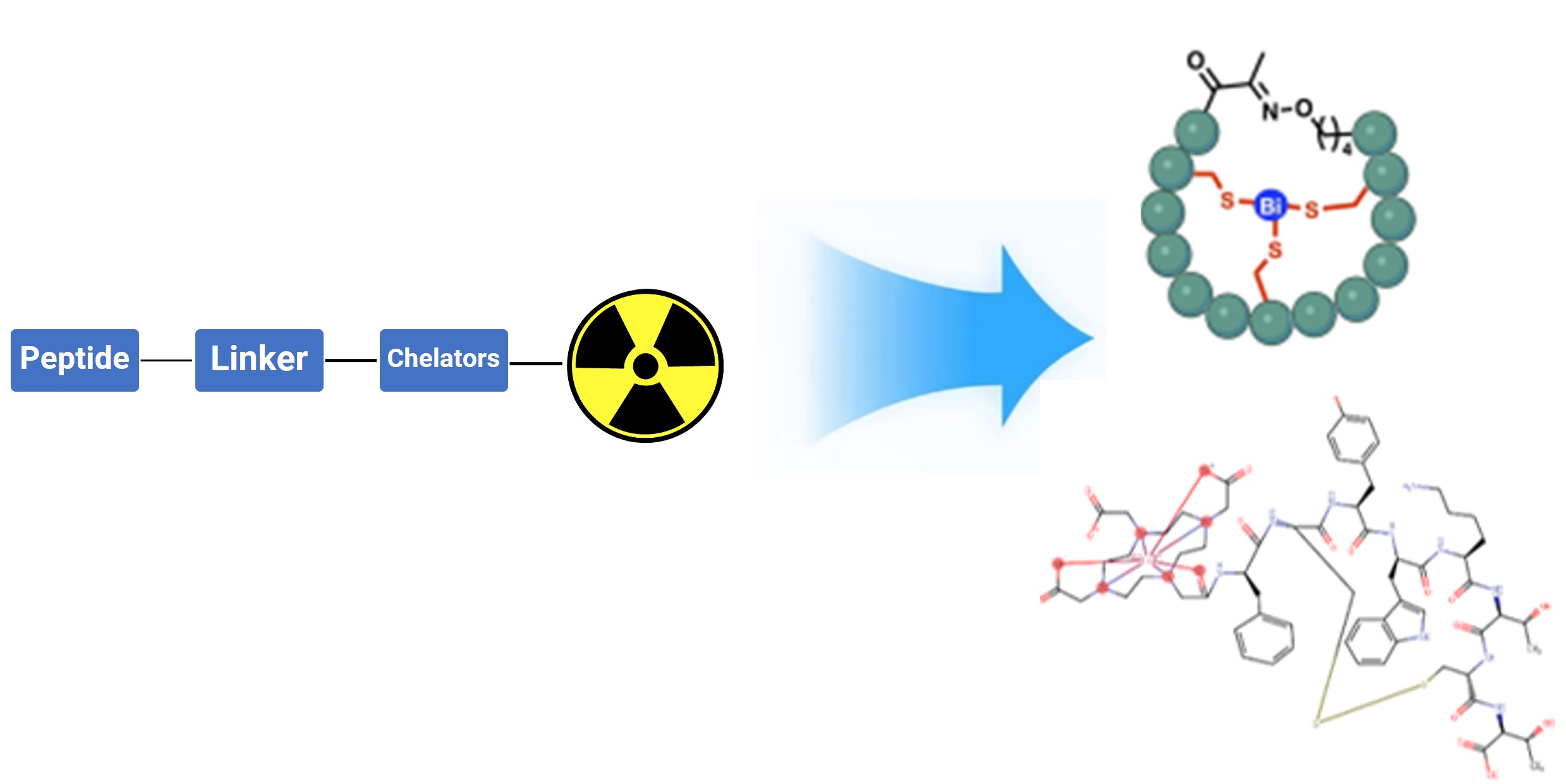
Figure 1. Structural example of a peptide receptor radionuclide therapies (PRRTs) or diagnostic PRCs
Radionuclides
Radionuclides exert therapeutic effects by damaging tumor cell DNA through the release of α, β, or γ rays during decay. Rays with stronger penetration capabilities are also utilized for diagnostic imaging. Currently, over 30 radionuclides are employed in disease diagnosis and treatment (see Table 2). 177Lu is the most common theranostic nuclide in PRRTs and diagnostic PRCs, capable of simultaneously emitting β-particles for therapy and γ-rays for imaging. Actinium-225 (225Ac) is a novel nuclide recently entering clinical use; it primarily releases α-particles, which have weaker tissue penetration but significantly higher lethality [1].
Table 2. Summary of characteristics of different radionuclides
Emitted Ray | Range in Tissue | DNA Damage | Characteristics & Applications | Representative Nuclides |
α-ray | 50–100 μm | Causes DNA double-strand breaks; highly destructive to cells, complex damage, difficult to repair. |
| Bismuth-213, Lead-212, Radium-223, Actinium-225, Thorium-227, Astatine-211 |
β-ray | 2–12 mm | Causes DNA single/double-strand breaks; damages DNA primarily through indirect free radical generation; relatively easy to repair. |
| Lutetium-177, Iodine-131, Yttrium-90, Copper-67, Rhenium-188 |
γ-ray | Low energy: mm Medium energy: cm | Causes DNA single/double-strand breaks; strong penetration can cause double-strand breaks at multiple sites; difficult to repair. |
| Lutetium-177, Gallium-67, Technetium-99m, Strontium-131, Iodine-125 |
Peptides
Peptides generally consist of 10 to 40 amino acids with a molecular weight typically under 10 kDa. Structurally, they can be classified into linear peptides composed of natural amino acids, linear peptides containing non-natural amino acids, and cyclic peptides.
Peptide drugs combine the advantages of small molecules and biologics:
High targeting and specificity: Peptides bind to specific receptors with high affinity, resulting in lower side effects. For example, radiolabeled bombesin analogs can precisely target the Gastrin-Releasing Peptide Receptor (GRPR) overexpressed in tumor cells for early diagnosis and treatment of prostate cancer.
Low immunogenicity and strong tissue penetration.
Favorable metabolic properties: Composed of amino acids, their metabolic byproducts are generally non-toxic.
Stability optimization: Linear peptides have flexible conformations and are prone to protease degradation. Cyclization strategies restrict conformational flexibility, enhance structural stability, reduce metabolic degradation by exopeptidases, lower the dissociation constant, and improve target affinity. Consequently, cyclic peptides are generally preferred as ligands for PRRTs and diagnostic PRCs to enhance in vivo stability and efficacy.
Chelators
The binding of the radionuclide to the ligand usually requires a chelator. The choice of chelator depends on the specific nuclide; common options include DOTA, DTPA, NOTA, and TETA.
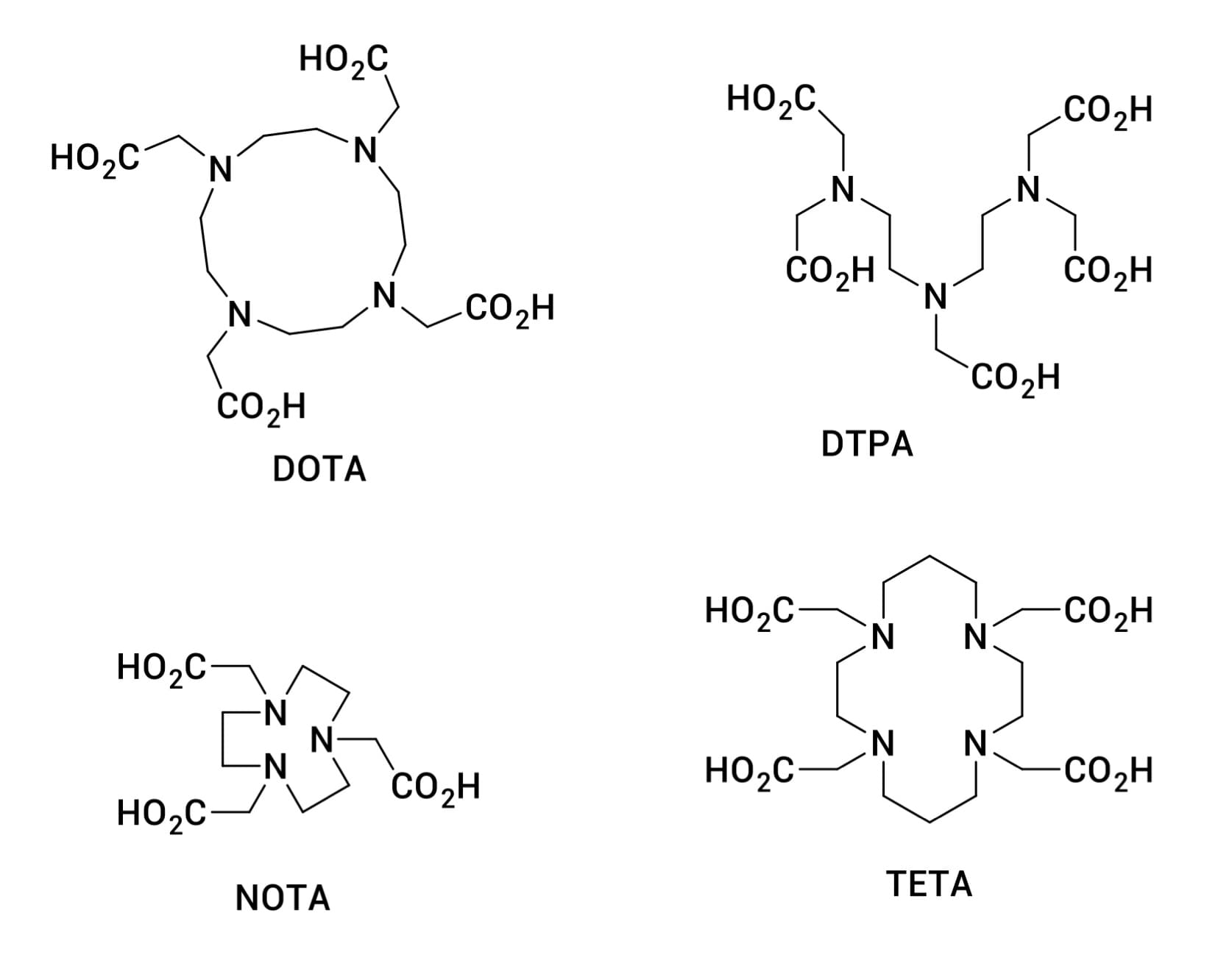
Figure 2. Schematic structures of common chelators
Mechanisms of Action and Key Advantages of PRRTs and Diagnostic PRCs
Mechanism of Action of PRRTs and Diagnostic PRCs
Precise targeting and theranostic integration are the core strengths of PRRTs and PRCs. They deliver radionuclides to the target lesion via the targeting peptide, inducing cell death through emitted rays or particles. Beyond direct cytotoxicity to target cells, PRRTs exert indirect killing effects on neighboring tumor cells through "cross-fire" and "bystander" effects. They may also inhibit distant cells by activating radiation-induced immune responses and the abscopal effect. Furthermore, strategies using the same targeting peptide loaded with different nuclides allow for a precise "diagnose first, treat later" theranostic workflow in clinical settings [1].
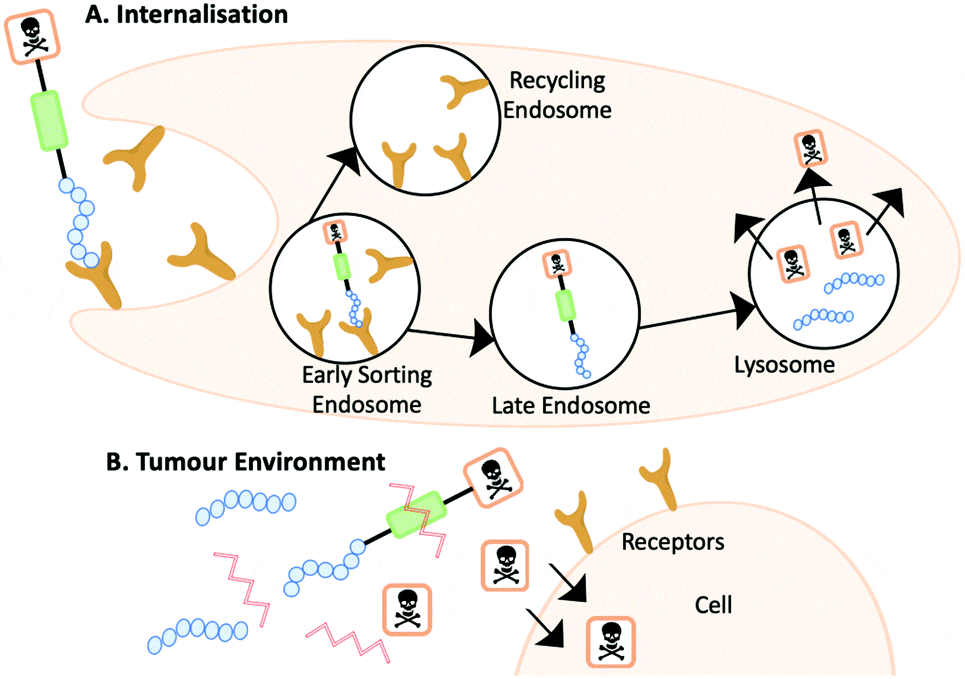
Figure 3. Schematic mechanism of action of PRRTs and PRCs [2]
Advantages of PRRTs and PRCs
PRCs stand out in the radiopharmaceutical landscape due to high target affinity, low immunogenicity, strong tissue penetration, and rapid blood clearance.
High Target Affinity
Given that PRRTs and PRCs drugs release radiation immediately upon entering the blood circulation, their targeting specificity must be exceptionally high. Christoph Nitsche's team [3] systematically compared two high-affinity peptides from bismuth and arsenic series (2b, 3a) against their linear analogs (2, 3). The study showed that the dissociation constants (KD) of the peptide-bismuth bicyclic compounds for streptavidin were in the 10 µM range—two orders of magnitude lower than their linear counterparts. This indicates that structural constraints can significantly enhance the affinity of PRRTs and PRCs for their targets, thereby improving efficacy.
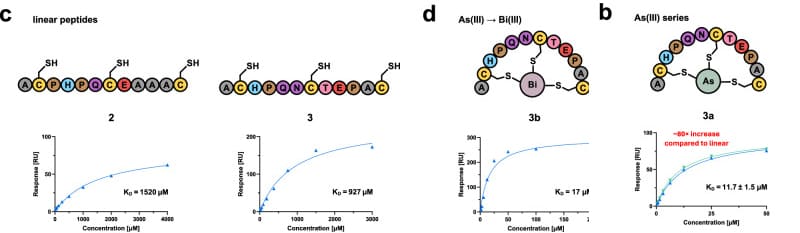
Figure 4. Comparison of dissociation constants for linear peptides (2, 3) and peptide-bismuth/arsenic bicycles (2b, 3a) [3]
Bicyclic peptides possess precise targeting specificity comparable to antibodies. For instance, the Bicycle® Radionuclide Conjugate (BRC) Pb-BCY20603 binds with high affinity to the tumor antigen MT1-MMP and carries Lead-212, an α-particle emitter. Preclinical studies demonstrated that 24 hours post-injection, radioactive uptake at the tumor site remained above 45% of the injected dose per gram (ID/g), showing excellent tumor targeting and significant anti-tumor activity in rodent xenograft models.
Strong Tissue Penetration
Antibodies, due to their large molecular weight and complex structure, are easily intercepted by vascular barriers in tumor tissues, reducing penetration efficiency. In contrast, peptides have lower molecular weights and can penetrate tissues rapidly and deeply, targeting lesions from the inside out. PRRTs leverage this advantage to accumulate quickly at the tumor site for highly efficient treatment. Structurally, cyclic peptides demonstrate superior cytosolic transport efficiency compared to linear peptides. Pb-BCY20603, utilizing bicyclic peptide technology, can penetrate the dense connective tissue barriers in solid tumors to reach the core, offering a significant advantage over traditional biologics.
High Clearance Rate
The characteristics of peptides endow PRRTs and PRCs with controllable pharmacokinetic half-lives and predominantly renal clearance pathways, avoiding the hepatic and gastrointestinal toxicities common in other drug modalities. The superior metabolic profile of PRRTs and PRCs is highly correlated with this renal dominance; they are rapidly cleared from the blood, and the short half-life helps reduce toxic side effects caused by off-target radiation. For example, 177Lu-DOTA-IBA clears quickly from the blood, with the portion not bound to bone primarily excreted via the urinary system. Four hours post-injection, the residual blood level is only 0.085 ± 0.011 % ID/g [4].
Metabolic Characteristics and Key Considerations for PRRT and PRC In Vitro and in vivo Studies
Metabolism of PRRTs and PRCs stems from both the radionuclide and the peptide. During metabolism, there is a risk of nuclide dissociation, which may cause radiation toxicity to normal tissues. Therefore, research must focus not only on the conjugate itself but also on the nuclide- dissociated peptide and its products. The chemical and biological properties of the peptide determine the in vivo biodistribution of the drug. Affinity and cell uptake are highly correlated with peptide integrity; enzymatic cleavage of the receptor-specific C-terminal sequence can lead to a complete loss of affinity and uptake. Thus, a deep understanding of metabolic mechanisms is critical for structural optimization and drug development [5].
Metabolic and Clearance Characteristics
Peptide metabolism is diverse and influenced by structure, molecular weight, and hydrophilicity. Mechanisms include peptidase hydrolysis, endocytic elimination, target-mediated elimination, and glomerular filtration, with enzymatic elimination and glomerular filtration being dominant. The kidney is the primary metabolic organ, and metabolites are excreted mainly via urine and feces [6].
Peptides are primarily metabolized by peptidases, divided into exopeptidases and endopeptidases, which are widely distributed in the liver, kidney, and gastrointestinal tract. Endopeptidases hydrolyze peptides into oligopeptides, which exopeptidases further break down into amino acids. Although the metabolic pathway is relatively singular (hydrolysis), the widespread distribution of hydrolases makes the system complex. Liver and kidney S9 fractions or homogenates are the most commonly used test systems.
Molecular configuration directly affects stability. Christoph Nitsche's team [7] studied the stability of linear, monocyclic, bicyclic, and tricyclic peptides in protease targets and human plasma. Results showed that linear and monocyclic peptides were prone to cleavage in protease target experiments, whereas tricyclic peptides maintained over 95% stability within 24 hours. In human plasma, bicyclic and tricyclic peptides showed enhanced stability (>65%) over 90 minutes. In summary, cyclic peptides exhibit superior stability in both protease targets (Figure 5C-D) and human plasma (Figure 5).
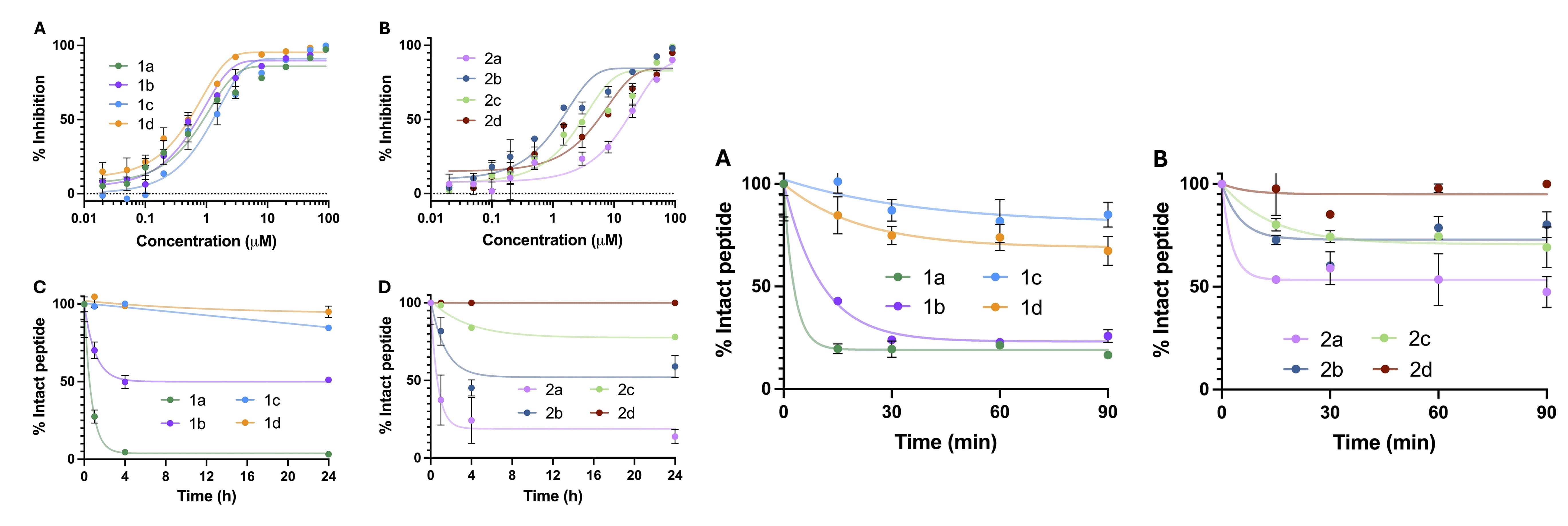
Figure 5. Stability comparison of linear (1a, 2a), monocyclic (1b, 2b), bicyclic (1c, 2c), and tricyclic (1d, 2d) peptides in protease targets and human plasma [7]
Voss Saan et al. [10] investigated the stability of peptide-bismuth bicycles in the presence of glutathione (GSH). Studies showed no hydrolysis within 48 hours at GSH concentrations up to 2 mM. However, at 10 mM GSH, 30% hydrolysis was observed. Another study [7] (Figure 6A-B) indicated that peptide-bismuth bicycle 2c showed slightly lower stability (>85% unchanged), while peptide-bismuth tricycle 2d maintained >95% integrity even in the presence of up to 25 equivalents of GSH. Therefore, different concentrations of GSH have different effects on the stability of polypeptides. Meanwhile, a given GSH concentration differentially influences polypeptides depending on their structural features (e.g., those in which nucleophilic residues and thiol groups are covalently linked).
The study also found that EDTA concentration significantly impacts stability. In the presence of the chelator EDTA, bismuth tends to bind with EDTA (Figure 6C-D). For both bicyclic and tricyclic peptides, the remaining PRC rate was below 80% under 5–25 equivalents of EDTA. Compared to bicyclic peptides, peptide-bismuth tricycles exhibited higher stability, though the effect of EDTA varies across different peptide-nuclide conjugate sequences.
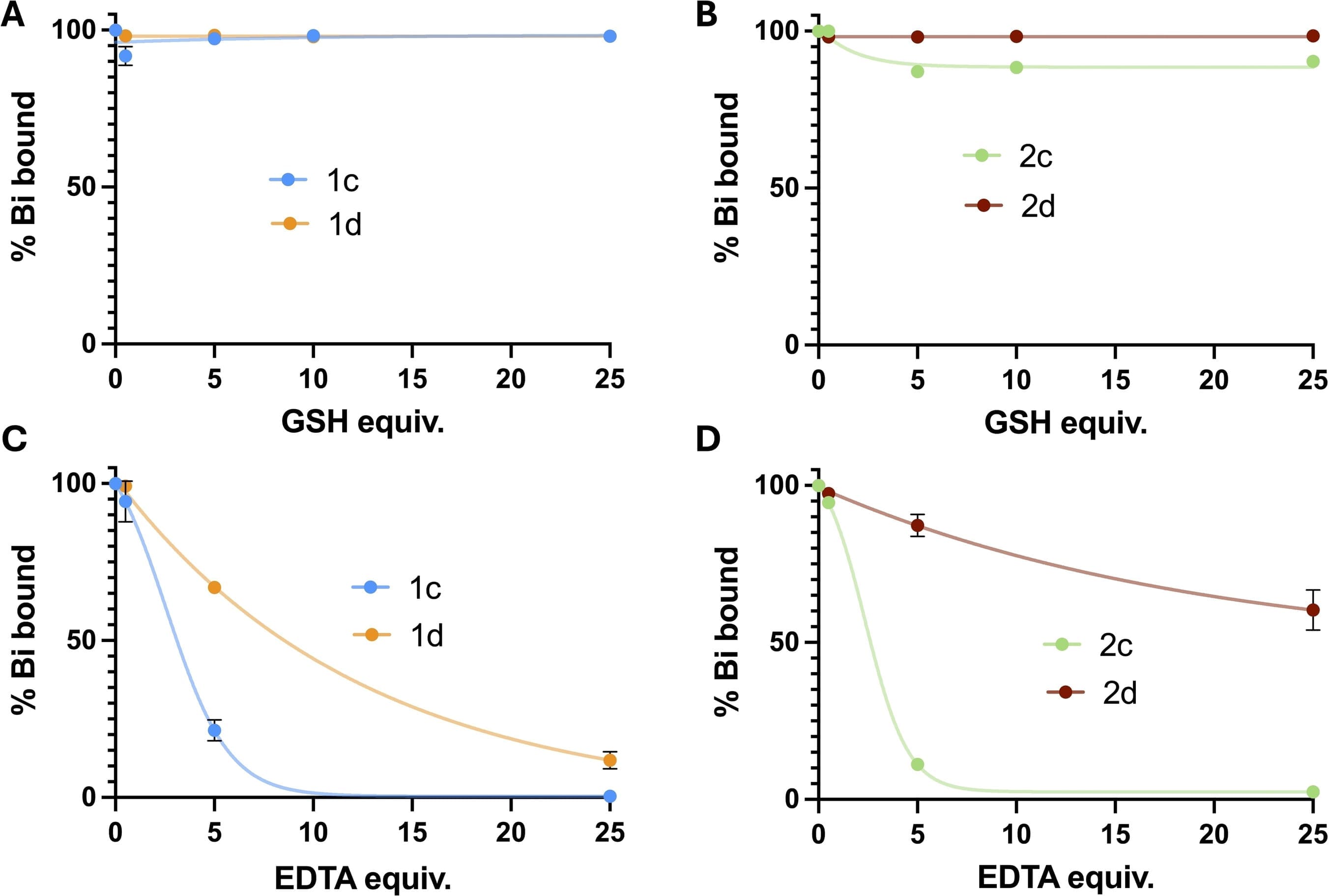
Figure 6. Stability of bicyclic (1c, 2c) and tricyclic (1d, 2d) peptides in GSH and EDTA [7]
Anton et al. [8] conducted in vitro human and in vivo mouse metabolic studies on a [177Lu]-labeled MG analog (DOTA-DGlu-Pro-Tyr-Gly-Trp-(N-Me)Nle-Asp-1Nal-NH2). Results indicated metabolic stability in human matrices followed the order: serum > liver homogenate > kidney homogenate. They also studied the stability of its four main metabolites ([177Lu]Lu-M1 to [177Lu]Lu-M4) after 24 hours of incubation in serum, finding that stability significantly decreased when protein binding of the PRC or metabolite was low, as high levels of free radiolabeled peptide are more susceptible to hydrolysis.
In vivo studies in mice showed that 60 minutes post-injection, the remaining PRC in whole blood was 56.9%, with four radiometabolites detected ([177Lu]Lu-M1–M3 and M5), accounting for 43.1%. In the liver at 60 minutes, the remaining PRC was 47.7%, with six metabolites (M1–M6) accounting for 82.0%. In the kidney and urine, identical metabolites were observed (except M7). Metabolites M1, M3, and M7 had low proportions (<10%), while the other four exceeded 10% with an increasing trend. The intact labeled MG analog accounted for only 7.0% in the kidney and 18% in the urine.
In conclusion, the labeled MG analog showed good stability in whole blood and liver but poor stability in the kidney, identifying the kidney as the primary clearance pathway. The seven metabolites detected in the liver and kidney were formed by C-terminal amide bond hydrolysis and sequential C-terminal peptide bond hydrolysis. However, no hydrolysis of the peptide bond between D-Glu and L-Pro was observed, suggesting the tertiary amide bond introduced into the peptide backbone may provide a stabilizing effect. Table 3 summarizes the proportions of different radiometabolites in mice.
Table 3. Proportions of radiometabolites in different matrices [8]
Peptide | Blood | Liver | Kidney | Urine | ||||
30 min | 60 min | 30 min | 60 min | 30 min | 60 min | 30 min | 60 min | |
[177Lu]Lu-1 | 64.1% | 56.9% | 53.9% | 47.7% | 18.0% | 7.0% | 19.4% | 18.0% |
[177Lu]Lu-M1 | 9.9% | 5.8% | 4.8% | 4.6% | 2.5% | - | 1.4% | - |
[177Lu]Lu-M2 | 3.8% | 7.3% | 7.2% | 6.9% | 20.7% | 12.3% | 40.5% | 40.3% |
[177Lu]Lu-M3 | 17.0% | 20.4% | 8.5% | 5.6% | 7.9% | - | 9.5% | 9.8% |
[177Lu]Lu-M4 | - | - | 2.9% | 2.6% | - | - | - | - |
[177Lu]Lu-M5 | 5.3% | 9.6% | 14.4% | 12.9% | 27.3% | 23.7% | 28.7% | 31.3% |
[177Lu]Lu-M6 | - | - | 8.3% | 19.7% | 23.6% | 53.2% | - | - |
[177Lu]Lu-M7 |
| - | - | - | - | 3.8% | - | - |
[177Lu]Lu-M8 | - | - | - | - | - | - | - | - |
(Note: Refer to original source for specific data values)
Considerations for In Vitro Metabolic Studies
Given the high barrier to entry for radioisotope experiments, if the selected radionuclide has prior clinical usage, preclinical research can be conducted using non-radioactive nuclide-conjugated analogs [9].
When designing a metabolic study for PRRTs and PRCs, multiple factors must be considered to ensure accurate assessment. Unlike standard peptide drugs, researchers must consider not only peptidase hydrolysis but also the cleavage of linkers and chelators.
Since nuclides function via radiation without direct cell contact, nuclide dissociation—and the subsequent toxicity risk—is a critical focus. Metal nuclides are typically linked via chelators; however, if stronger chelators exist in the body, the metal may be captured, reducing efficacy. Glutathione (GSH) and Ethylenediaminetetraacetic acid (EDTA) are two vital considerations here.
Plasma is the preferred matrix for in vitro and in vivo metabolism studies due to accessibility and rich peptidase content. During plasma preparation, anticoagulants are necessary. Common types include EDTA and Heparin Sodium. Crucially, EDTA may affect enzyme activity or the stability of the nuclide within the PRRTs and PRCs, potentially skewing results. Therefore, experimental designs should prioritize Heparin Sodium to ensure data reliability.
When the radionuclide in a PRC is bound to sulfur atoms in the peptide, the effect of GSH concentration on metabolism requires further evaluation. Studies indicate that competition from thiol-containing compounds can impact the binding of peptide thiols to bismuth. To better assess in vivo stability, incubation systems containing GSH (such as liver S9, homogenates, or hepatocytes) may be more suitable for researching PRC metabolism.
Final Thoughts
As the global population ages and cancer incidence rises, pharmaceutical companies are aggressively positioning themselves in the nuclear medicine sector, driving rapid market growth. Peptide receptor radionuclide therapies (PRRTs) and diagnostic PRCs, as emerging precision theranostic agents, have become a key direction in nuclear medicine R&D due to their high affinity, low immunogenicity, excellent tissue penetration, and rapid clearance.
However, PRCs still face risks during metabolism, such as peptide hydrolysis and nuclide dissociation, which may cause radiation damage to non-target organs. Consequently, systematic metabolite identification is essential to fully characterize hydrolysis and degradation pathway, providing a reference for efficacy and toxicity studies. To meet this need, WuXi AppTec DMPK has established a comprehensive PRRT and diagnostic PRC metabolite identification platform. Centered on UPLC-UV-HRMS technology and combined with specialized data processing software, this platform provides reliable support for the analysis and optimization of PRRTs and PRCs metabolic behavior, facilitating the efficient development of precision oncology treatments.
Authors: Bingqian Guo, Ruixing Li, Weiqun Cao
Talk to a WuXi AppTec expert today to get the support you need to achieve your drug development goals.
Committed to accelerating drug discovery and development, we offer a full range of discovery screening, preclinical development, clinical drug metabolism, and pharmacokinetic (DMPK) platforms and services. With research facilities in the United States (New Jersey) and China (Shanghai, Suzhou, Nanjing, and Nantong), 1,000+ scientists, and over fifteen years of experience in Investigational New Drug (IND) application, our DMPK team at WuXi AppTec are serving 1,600+ global clients, and have successfully supported 1,700+ IND applications.
Reference
[1] Technical Guidelines for Clinical Evaluation of Radioactive In Vivo Therapeutic Drugs (2023).
[2] Bethany M. Cooper, et al. Peptides as a platform for targeted therapeutics for cancer: peptide–drug conjugates (PDCs). Chem. Soc. Rev, 2021, 50: 1480-1494.
[3] Sven Ullrich, et al. Phage-encoded bismuth bicycles enable instant access to targeted bioactive peptides. Communications Chemistry, 2024, 7: 143.
[4] Hongmei Li, et al. Biodistribution and dosimetry of 177Lu-DOTA-IBA for therapy of bone metastases. EJNMMI Research, 2024, 14: 30.
[5] Technical Guidelines for Clinical Evaluation of Radioactive In Vivo Diagnostic Drugs (2020).
[6] Yao Jinfeng. Progress in Metabolic Research of Peptide Drugs [J]. Chinese Pharmacological Bulletin, 2013, 29(7):5.
[7] Lani J. Davies, et al. Peptide–Bismuth Tricycles: Maximizing Stability by Constraint. Chem. Eur. J, 2025, 31: e202500064.
[8] Anton Amadeus Hörmann, et al. Initial In Vitro and In Vivo Evaluation of a Novel CCK2R Targeting Peptide Analog Labeled with Lutetium-177. Molecules, 2020, 25, 4585.
[9] Technical Guidelines for Non-clinical Evaluation of Radioactive In Vivo Diagnostic Drugs (2021).
[10] Voss Saan, et al. Cell-Penetrating Peptide-Bismuth Bicycles. Angew. Chem. Int. Ed, 2024, 63, e202318615.
Related Services and Platforms




-

 MetID (Metabolite Profiling and Identification)Learn More
MetID (Metabolite Profiling and Identification)Learn More -

 Novel Drug Modalities DMPK Enabling PlatformsLearn More
Novel Drug Modalities DMPK Enabling PlatformsLearn More -

 In Vitro MetID (Metabolite Profiling and Identification)Learn More
In Vitro MetID (Metabolite Profiling and Identification)Learn More -

 In Vivo MetID (Metabolite Profiling and Identification)Learn More
In Vivo MetID (Metabolite Profiling and Identification)Learn More -

 Metabolite Biosynthesis and Structural CharacterizationLearn More
Metabolite Biosynthesis and Structural CharacterizationLearn More -

 Metabolites in Safety Testing (MIST)Learn More
Metabolites in Safety Testing (MIST)Learn More -

 PROTAC DMPK ServicesLearn More
PROTAC DMPK ServicesLearn More -

 ADC DMPK ServicesLearn More
ADC DMPK ServicesLearn More -

 Oligo DMPK ServicesLearn More
Oligo DMPK ServicesLearn More -

 PDC DMPK ServicesLearn More
PDC DMPK ServicesLearn More -

 Peptide DMPK ServicesLearn More
Peptide DMPK ServicesLearn More -

 mRNA DMPK ServicesLearn More
mRNA DMPK ServicesLearn More -

 Covalent Drugs DMPK ServicesLearn More
Covalent Drugs DMPK ServicesLearn More
Stay Connected
Keep up with the latest news and insights.